Drug Watch
Latest News

Latest Videos

Shorts





CME Content
More News

Unlike approved AD biologics and JAK inhibitors targeting specific cytokine pathways, GX-03 is designed to modulate inflammatory signaling locally within the skin microenvironment.

Every week, we cut through the noise to bring clinicians the trial results, approvals, and emerging therapies that are actually moving the needle.

Apogee plans to initiate 3 phase 3 trials of zumilokibart in moderate to severe atopic dermatitis—ADventure 1, ADventure 2, and ADventure TCS—in the second half of 2026.
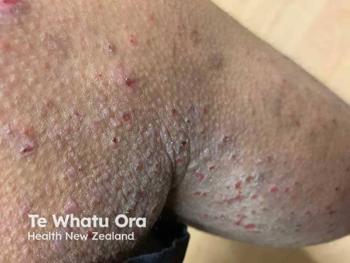
Among more than 1700 patients enrolled in the long-term extension study, the majority transitioned from a moderate to severe AD phenotype at baseline to a mild phenotype within 12 weeks, with that shift maintained through 2 years.

The fixed-dose adapalene 0.1% and benzoyl peroxide 2.5% topical OTC gel broadens nonprescription access to a combination topical regimen that has been used as a prescription product for more than 15 years.

Phase 3 data presented at AAD 2026 showed that amlitelimab significantly improved skin clearance and disease severity in patients aged 12 years and older with moderate to severe atopic dermatitis.
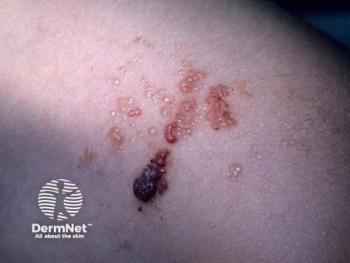
Phase 3 SELVA and phase 2 TOIVA data show Qtorin rapamycin met primary end points for microcystic lymphatic and cutaneous venous malformations.
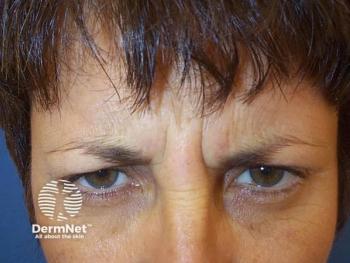
Data presented at SCALE 2026 showed that Ipsen’s investigational recombinant neuroinhibitor achieved significant and sustained improvement in moderate to severe glabellar lines.

Every week, we cut through the noise to bring clinicians the trial results, approvals, and emerging therapies that are actually moving the needle.
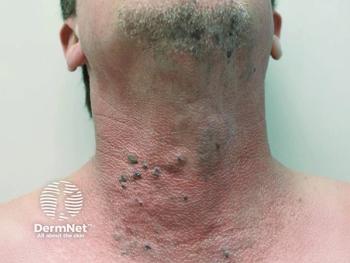
Phase 2 TOIVA data show Palvella's QTORIN rapamycin reduces bleeding and improves quality of life in cutaneous venous malformations, addressing an unmet treatment need.

Every week, we cut through the noise to bring clinicians the trial results, approvals, and emerging therapies that are actually moving the needle.
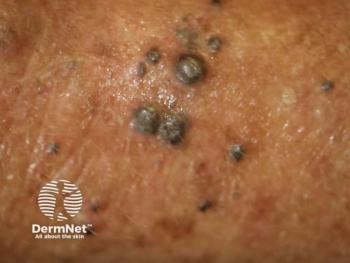
A phase 1/2 trial enrolling patients with refractory melanoma is now actively recruiting.
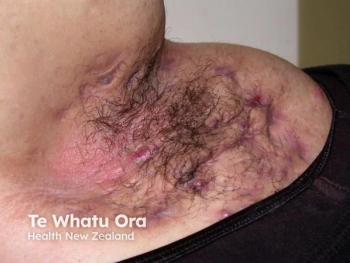
Initial phase 2 data show abdakibart (AVTX-009) boosts HiSCR responses in moderate to severe HS, with favorable safety, paving way for phase 3.

Every week, we cut through the noise to bring clinicians the trial results, approvals, and emerging therapies that are actually moving the needle.

Review the latest FDA pediatric dermatology updates, including dupilumab for childhood CSU, secukinumab for teen HS, and new CHE and infant eczema topical filings.

Maria Hordinsky, MD, breaks down phase 3 data that show topical clascoterone boosts male pattern hair growth for 12 months, targeting follicles without systemic hormonal adverse effects.
Aclaris Therapeutics shares promising Phase 1a data for ATI-052, showing long-acting dual Th2 blockade, and advances ATI-2138 toward Phase 2b lichen planus studies.

The application is based on phase 3 UP-AA trial data showing significant hair regrowth in patients with severe alopecia areata.

Interim phase 2a data show ORKA-001, a half-life extended IL-23 inhibitor, achieved complete skin clearance in 63.5% of patients at week 16.

Every week, we cut through the noise to bring clinicians the trial results, approvals, and emerging therapies that are actually moving the needle.
Potential FDA approval of TrenibotE was delayed due to manufacturing-related issues, with no concerns regarding safety, efficacy, or clinical trial data.
Early clinical trials indicate that dual- and triple-target antibodies may provide faster onset and more consistent itch relief in moderate to severe AD.
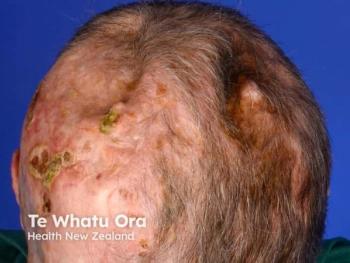
Medicus seeks FDA orphan drug status for SkinJect, a dissolvable microneedle doxorubicin patch targeting Gorlin-related basal cell carcinomas, aiming to reduce the need for repeated surgery.

The biologic is the first of its kind approved for young children with antihistamine-refractory CSU, expanding an existing indication that previously covered adults and adolescents.
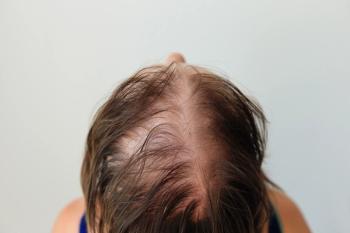
REZPEG shows growing hair regrowth at 52 weeks in severe alopecia areata, with favorable safety, hinting at a new biologic alternative to JAKs.