
VYNE Therapeutics updates on VYN202, revealing promising early results in psoriasis treatment despite a clinical hold due to safety concerns.

VYNE Therapeutics updates on VYN202, revealing promising early results in psoriasis treatment despite a clinical hold due to safety concerns.

A new paper highlights Alphyn Biologics' Zabalafin Hydrogel as a promising multitarget therapy for atopic dermatitis, addressing inflammation, itch, and bacterial complications.

Quoin Pharmaceuticals advances QRX003 for Netherton Syndrome, receiving FDA's RPD Designation and showing promising clinical results.

Nektar Therapeutics reveals positive results from the REZOLVE-AD study, showcasing rezpegaldesleukin's efficacy in treating moderate to severe AD.

Galderma launches clinical trials for nemolizumab, targeting systemic sclerosis and chronic pruritus of unknown origin, addressing critical patient needs.

The new PDUFA date is set for September 2025.

Dupilumab gains FDA approval as the first targeted therapy for bullous pemphigoid, offering hope for effective treatment and improved patient outcomes.

Pediatric dermatologist Mercedes E. Gonzalez, MD, discusses roflumilast's promise as a safe, nonsteroidal treatment for atopic dermatitis in infants.
Mercedes Gonzalez, MD, discusses the promising INTEGUMENT-INFANT trial, exploring a new non-steroidal treatment for infants with atopic dermatitis.

At the 2025 EULAR meeting, UCB presented new 3-year data on bimekizumab for PsA.
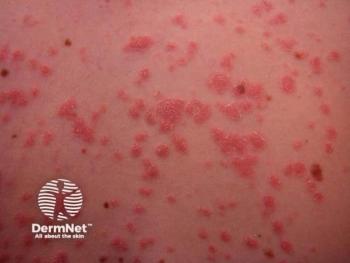
Zasocitinib emerges as a promising oral therapy for psoriasis and psoriatic arthritis, showcasing superior selectivity and safety compared to traditional JAK inhibitors.
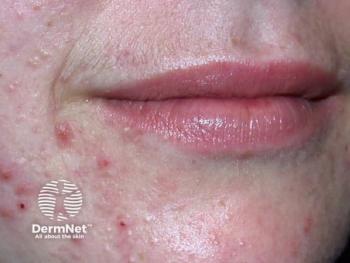
Sagimet Biosciences reveals denifanstat's promising phase 3 results, offering a novel oral treatment for moderate to severe acne with significant efficacy.

FDA approves Arcutis' roflumilast foam, a groundbreaking treatment for plaque psoriasis, offering a steroid-free, effective solution for patients aged 12 and older.

Quoin Pharmaceuticals advances QRX003 for Netherton Syndrome, receiving FDA clearance for a pivotal trial to assess its effectiveness and safety.

Accropeutics reveals promising Phase 2 trial results for AC-201, a selective TYK2/JAK1 inhibitor, showing significant efficacy in treating plaque psoriasis.

Albert Chiou, MD, MBA, discusses early clinical data on soquelitinib for atopic dermatitis, highlighting its targeted ITK inhibition, dose-dependent efficacy, favorable safety profile, and more.

Scott Batty, MD, and Chris VanDeusen, PhD, provided insights on AX-158's phase 2a results, presented at the SID Annual Meeting.

The promising safety and efficacy results were presented at the Society for Investigative Dermatology 2025 Annual Meeting.

Selective ITK inhibition with oral soquelitinib led to early and durable clinical responses, cytokine reduction, and increased T regulatory cells.

The FDA approved pz-cel, a groundbreaking gene therapy for recessive dystrophic epidermolysis bullosa, transforming treatment options for patients.

Dupilumab becomes the first targeted therapy approved for chronic spontaneous urticaria in more than a decade.

Having multiple therapeutic options would allow clinicians to tailor treatment and offer hope to patients who don't respond initially.
While approved for vitiligo and atopic dermatitis, ruxolitinib cream may help treat other inflammatory skin diseases.

Exclusive Q1 Report: A comprehensive look at the latest FDA approvals and pipeline developments in dermatology from the first quarter of 2025.

The novel treatments will be for patients with orphan conditions like Netherton Syndrome, Peeling Skin Syndrome, SAM Syndrome, and palmoplantar keratoderma.