Pediatric Dermatology
Latest News
Latest Videos

Shorts
CME Content
More News

Arcutis advances pediatric atopic dermatitis treatment with roflumilast cream, completing enrollment in key INTEGUMENT-INFANT trial.

Arcutis has expanded roflumilast cream in the US, a steroid-free treatment for mild to moderate AD in young children.
The FDA has approved roflumilast 0.05% cream for atopic dermatitis in children aged 2 years and older, extending its use to a critical pediatric population.

Berdazimer shows promise for treating MC in pediatric patients, even in those with a history of atopic dermatitis, according to a poster at Maui Derm NP+PA Fall.

Nicole Harter, MD, explores ruxolitinib 1.5% cream for pediatric atopic dermatitis, offering a novel, effective treatment option for children aged 2 to 11.

Ruxolitinib cream has become the first topical JAK inhibitor available for pediatric atopic dermatitis (AD) in the US.

Experts discuss innovative topical therapies for pediatric AD, emphasizing individualized treatment and the importance of managing itch and sleep quality.
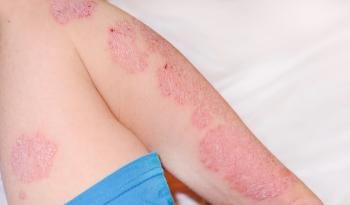
Arcutis Biotherapeutics seeks FDA approval for Zoryve cream to treat plaque psoriasis in children as young as 2, addressing a critical treatment gap.

Lisa Swanson, MD, FAAD, shares innovative insights on pediatric dermatology, emphasizing effective treatments and communication strategies for young patients at DERM 2025.

Explore common pediatric summer skin issues, from rashes to sun sensitivity, and learn effective prevention and management strategies for healthy skin.

Walter Liszewski, MD, shares expert strategies for managing body surface area limitations in AD treatment using non-steroidal topicals, and explored key unmet needs in the current therapeutic landscape.

At the Elevate-Derm Summer 2025 meeting, Walter Liszewksi, MD, discussed how new topical therapies, particularly JAK inhibitors, are transforming AD care with rapid itch relief, effectiveness in complex cases, and patient-friendly formulations for sensitive areas.

Pediatric psoriasis poses unique treatment challenges, highlighting the importance of early intervention and awareness of comorbidities in young patients.

Walter Liszewski, MD, discusses the personalized and evolving treatment landscape for moderate to severe AD, emphasizing a tailored approach using new biologics and non-steroidal topicals to meet individual patient needs and expectations.

Tina Bhutani, MD, MAS, highlights emerging topical treatments for pediatric atopic dermatitis, emphasizing improved adherence in younger children, the potential impact of ruxolitinib FDA approval for ages 2 and up, and the promise of ongoing trials in infants under 2 years.

Tina Bhutani, MD, MAS, discusses innovative pediatric atopic dermatitis treatments, including ruxolitinib cream, emphasizing collaborative decision-making for effective management.
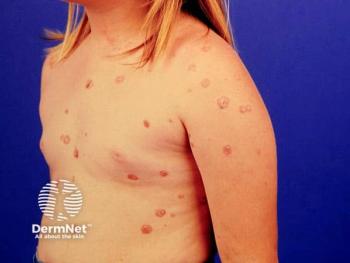
Parents advocate for children with chronic skin conditions, highlighting school challenges and the need for better support in pediatric dermatology.

Shanna Miranti, MPAS, PA-C, discusses the impact of sleep loss in pediatric atopic dermatitis and the potential of emerging therapies in young patients, such as ruxolitinib cream, to improve outcomes.

In a discussion on pediatric AD management, Shanna Miranti, MPAS, PA-C, emphasized the enduring role of topical treatments for residual itch and flares, and highlighted the importance of rapid-acting agents such as ruxolitinib cream.

Coates addresses social determinants of health in pediatric dermatology, emphasizing their impact on treatment and patient outcomes.

Shanna Miranti, MPAS, PA-C, describes her clinical approach for managing moderate to severe pediatric atopic dermatitis and how she selects between the 3 available non-steroidal topicals.

The pediatric dermatology community mourns the loss of Dr Robert Sidbury, a beloved leader and mentor, celebrated for his compassion and groundbreaking contributions.

Eichenfield discussed the ever-evolving armamentarium, and how to navigate changes in the field.

Traditional mouse models for EBS are limited, prompting Simpson's lab to create more accurate human-based tissue models.

At Elevate-Derm Summer, Jim Treat, MD, outlined diagnostic limitations of imaging and the importance of recognizing capillary malformation patterns linked to other diseases.