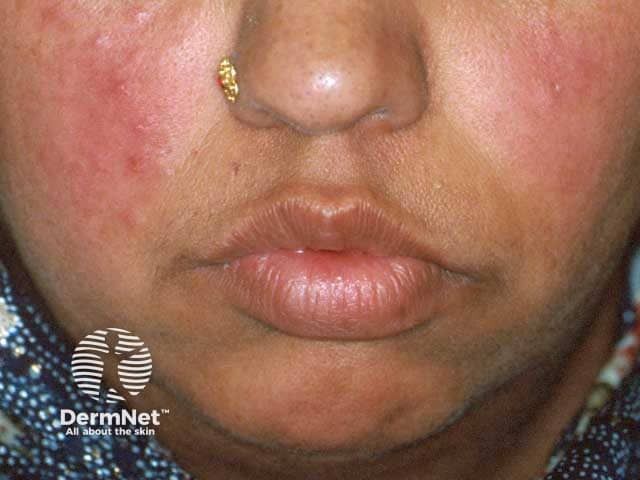
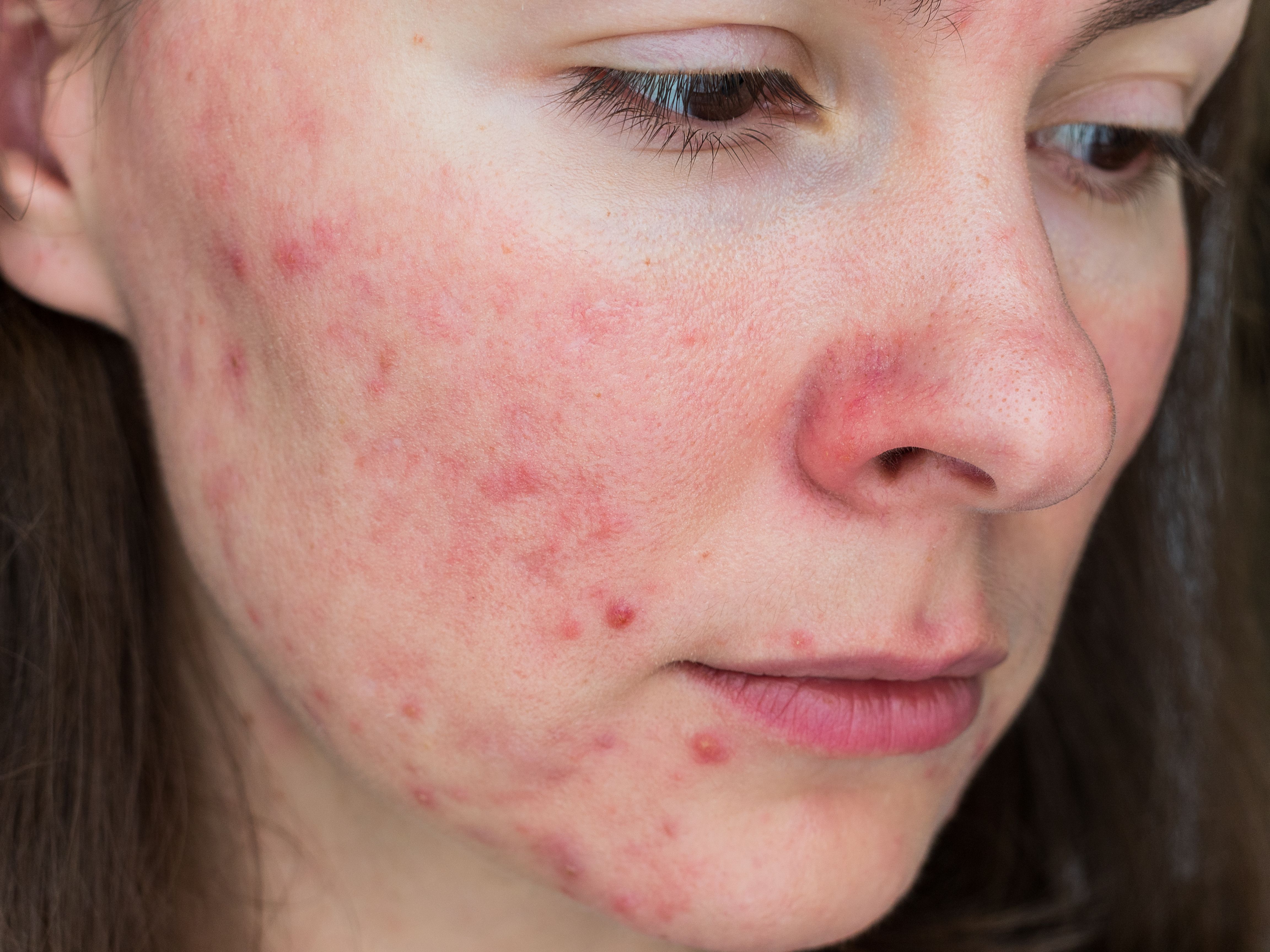
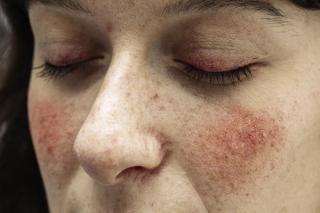
Rosacea
Latest News
Latest Videos

CME Content
More News

IKI Skin Care by Maria Urbani, MD offers effective solutions for rosacea and sensitive skin through science-backed, minimalist formulations.
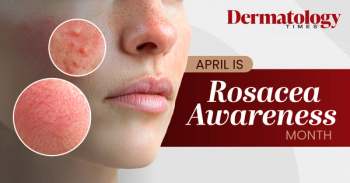
Explore the latest insights, strategies, and treatments for patients with rosacea as Dermatology Times looks back on Rosacea Awareness Month.
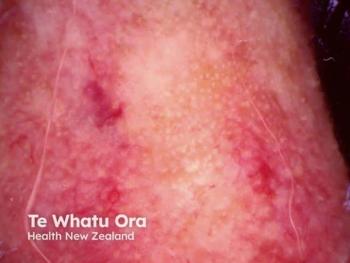
Discover innovative rosacea treatments combining injectable hyaluronic acid with traditional therapies to enhance skin health and boost patient confidence.
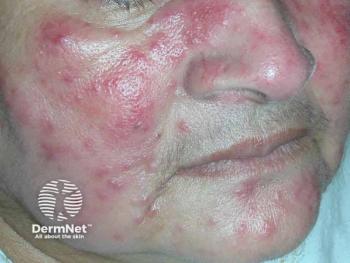
Researchers conduct a review to understand the efficacy and safety of topical minocycline for the treatment of moderate to severe papulopustular rosacea.
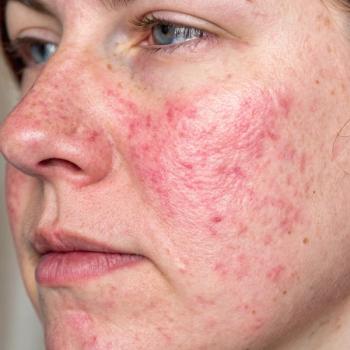
Over the past few years, research on the microbiome and dysbiosis has gained interest in the potential exacerbation of several skin diseases, including rosacea.

Improvements in hydration, erythema, itching, and burning were observed after 12 weeks of treatment for papulopustular rosacea.

Experts explore how rosacea research is evolving, with new treatments, gaps in care, and a push for more inclusive, targeted approaches.

The product was safe and effective after 4 weeks of topical use, specifically on patients with darker skin types.

Researchers behind a recent review shared expert insights, treatment guidelines, and differential diagnosis techniques for pediatric rosacea.

Each treatment had benefits and disadvantages for patients who struggle with rosacea-associated erythema.

The lowest-dose oral minocycline for rosacea is now available by prescription following FDA approval in November 2024.

The findings of the MVOR-1 and MVOR-2 studies are published in JAMA Dermatology, supporting the rosacea drug’s recent FDA approval.

Patients receiving SSA treatments showed significant improvements after just 2 sessions, supporting its rapid efficacy.
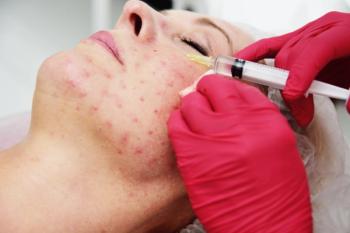
Symptoms of rosacea and dependence on oral medication were reduced in all 5 case studies.

Researchers found the combination therapy significantly reduced rosacea severity, with 64% achieving mild or normal status in just 4 weeks.
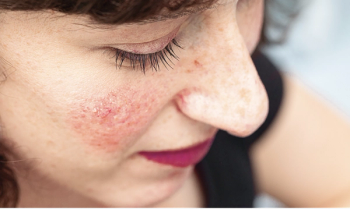
DFD-29, FDA-approved in 2024, revolutionizes rosacea management by addressing both erythema and inflammatory lesions with a novel extended-release formulation.

As 2024 comes to a close, Dermatology Times is taking a look back at the studies, therapies, and advances in rosacea this year.
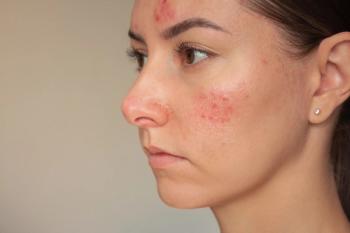
In Middle Eastern communities specifically, patients with higher disease severity and duration are predicted to have poorer psychological outcomes.

At the 2024 SDPA Fall Conference, Hilary Baldwin, MD, highlighted innovative strategies for managing complex rosacea cases, the importance of combination approaches, and a slowing treatment pipeline.

The minocycline hydrochloride extended release capsules (40 mg) represent a new standard of care for patients with rosacea.

Designed to fill a gap in rosacea evaluation, the TRoSA scale provides an objective method for tracking telangiectasia severity, which could also be integrated with AI for automated assessments.

The trial results indicated improved disease control, integrity of skin barrier, and minimal reactions for patients with rosacea.

Experts shared pearls for acne and rosacea care at Maui Derm NP+PA Fall.

Hilary Baldwin, MD, emphasized involving patients in the decision-making process to create a personalized and cost-effective plan.

Del Rosso’s cases emphasized the importance of thorough patient evaluation and communication with other health care providers.