Drug Watch
Latest News
Latest Videos

CME Content
More News

Disc Medicine filed an NDA for bitopertin to treat erythropoietic protoporphyria (EPP) and X-linked protoporphyria (XLP) in patients ≥12 years.
The vaccine is designed to stimulate T cell responses against IDO1- and PD-L1–positive cells within the tumor microenvironment.
While VELA-1 achieved statistical significance under all analyses, VELA-2 narrowly missed its composite primary endpoint due to higher-than-expected placebo outcomes.

Fewer than 20% of eligible patients currently receive injectable therapies, highlighting the need for oral alternatives.

Phase 3 PROTOSTAR data support new indications, expanding treatment options for children impacted by chronic immune-mediated diseases.

Bambusa Therapeutics reveals promising phase I trial results for BBT001, targeting atopic dermatitis with dual-action bispecific antibodies.

TRIV-509 is a dual KLK5/7 inhibitor that aims to address barrier dysfunction, inflammation, and itch in moderate to severe AD.

Ruxolitinib cream has become the first topical JAK inhibitor available for pediatric atopic dermatitis (AD) in the US.
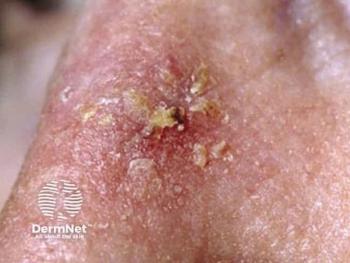
Biofrontera advances closer to expanding the Ameluz PDT label, as phase 3 follow-up data on actinic keratoses are expected next year.

The FDA's groundbreaking decision to waive clinical efficacy studies for a monoclonal antibody biosimilar revolutionizes drug approval, reducing costs and enhancing patient access.

Phase 3 trials demonstrated that icotrokinra met all primary and co-primary endpoints, including PASI 90 and IGA 0/1 responses.

The expanded voluntary recall is due to the potential microbial contamination of Burkholderia cepacia complex.

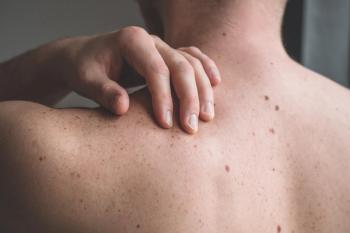
Amlitelimab shows promising phase 3 results in treating atopic dermatitis, achieving significant skin clearance and potential for infrequent dosing.
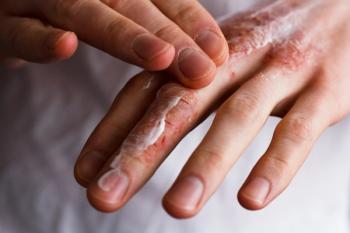
LEO Pharma's delgocitinib cream for chronic hand eczema is now available to prescribe in the US.

The FDA cited concerns about patient population heterogeneity and confirmatory trial design.
Nektar Therapeutics and Jonathan Zalevsky, PhD, are exploring REZPEG's potential in treating alopecia, atopic dermatitis, lupus, and diabetes, aiming for groundbreaking autoimmune therapies.

Nektar Therapeutics explores rezpegaldesleukin's potential as a groundbreaking treatment for alopecia areata, aiming for FDA approval and improved patient outcomes.
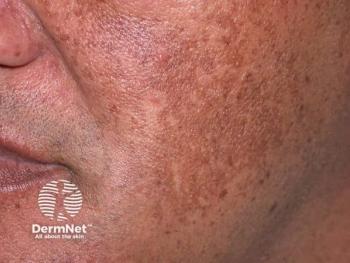
Patients receiving BoNT-A plus topical cream reported greater satisfaction and better skin lightness than with cream alone.
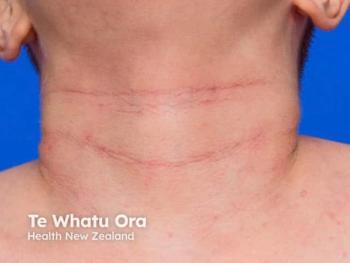
Amlitelimab shows promise in enhancing clinical outcomes and patient quality of life in atopic dermatitis, offering potential for long-term disease control.
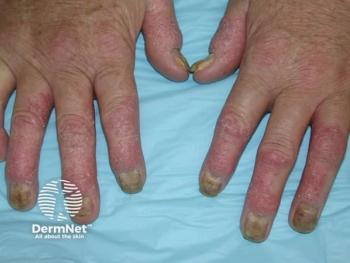
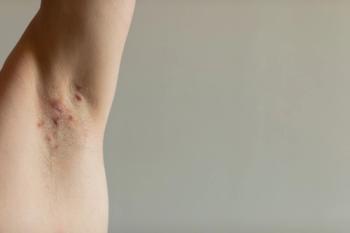
Deucravacitinib-treated patients achieved superior clinical responses compared to placebo at week 16 in both trials.
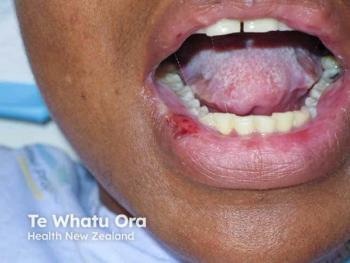
The open-label trial showed dusquetide produced similar results to apremilast in reducing oral ulcers, pain, and ulcer duration.

PC111, a novel therapy from Scinai, targets pemphigus vulgaris and SJS/TEN, offering rapid relief without immunosuppression.

VYNE Therapeutics reveals disappointing phase 2b trial results for repibresib gel in nonsegmental vitiligo, missing key efficacy endpoints.

Nektar Therapeutics advances alopecia areata treatment with FDA's Fast Track designation for rezpegaldesleukin, targeting immune modulation in chronic hair loss.

The approval makes delgocitinib cream the first and only approved therapy in the US for CHE in adults.