Drug Watch
Latest News
Latest Videos

CME Content
More News
FDA will review LEO Pharma's delgocitinib cream for adolescent patients aged 12-17 with CHE, backed by phase 3 gains and addressing a major treatment gap.

Every week, we cut through the noise to bring clinicians the trial results, approvals, and emerging therapies that are actually moving the needle.

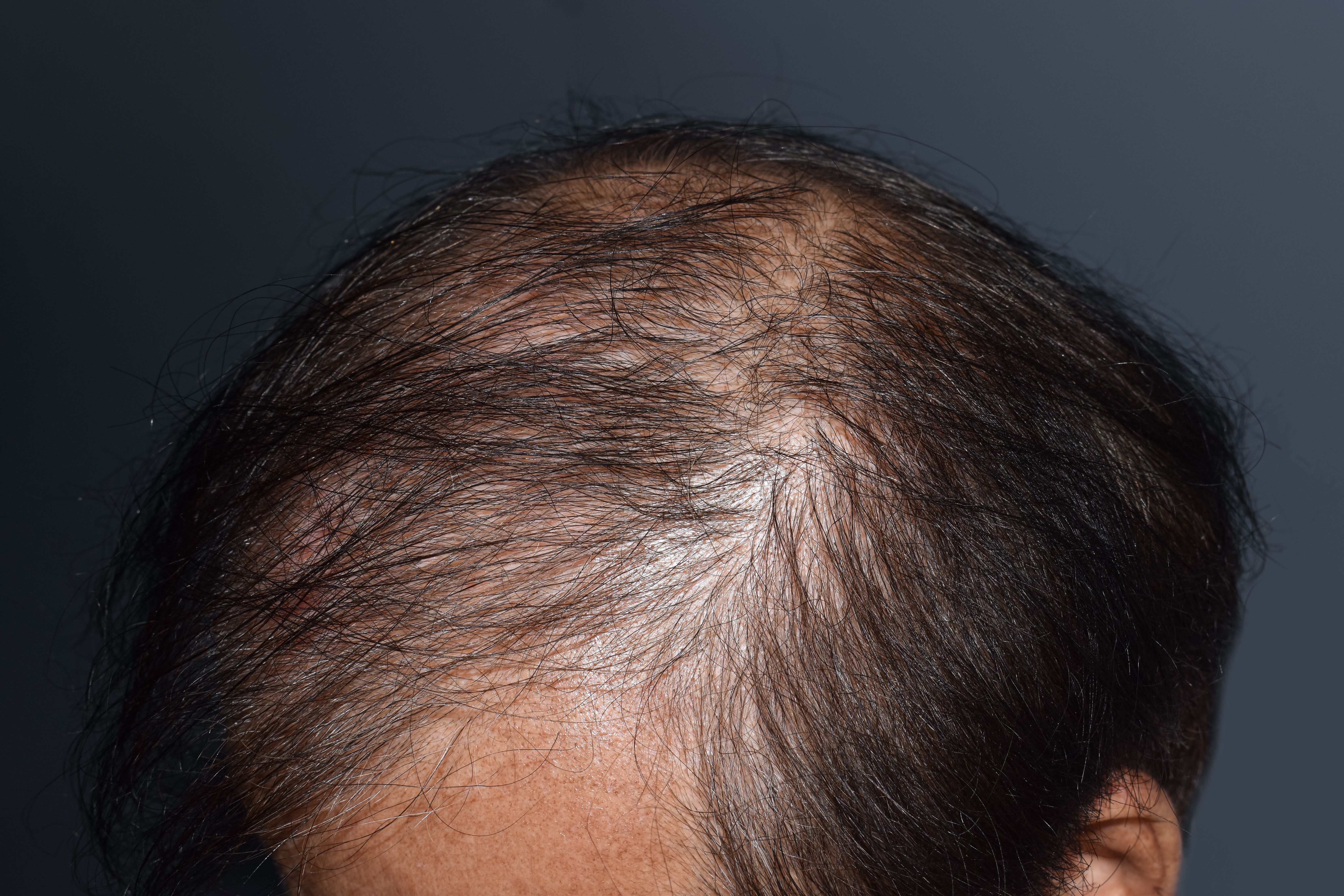
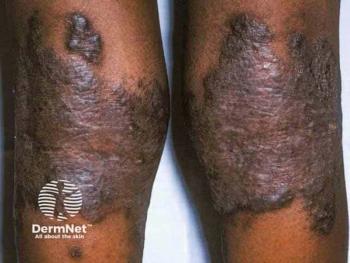
Phase 2/3 brepocitinib trial will target moderate to severe LPP, aiming to calm inflammation and symptoms and potentially deliver the first FDA-approved systemic option.

Every week, we cut through the noise to bring clinicians the trial results, approvals, and emerging therapies that are actually moving the needle.

Mark Jackson, MD, of Evommune, provides a therapeutic update on the investigational atopic dermatitis drug, citing strong efficacy and safety after just 2 doses, with phase 2b research on the way.

Kimball presented new data on MoonLake Immunotherapeutics’ sonelokimab and reflected on other HS updates presented at AAD 2026.
Hoth Therapeutics has received regulatory approval in Hungary to expand the phase 2 trial into Europe, with Spain and Poland approvals anticipated.
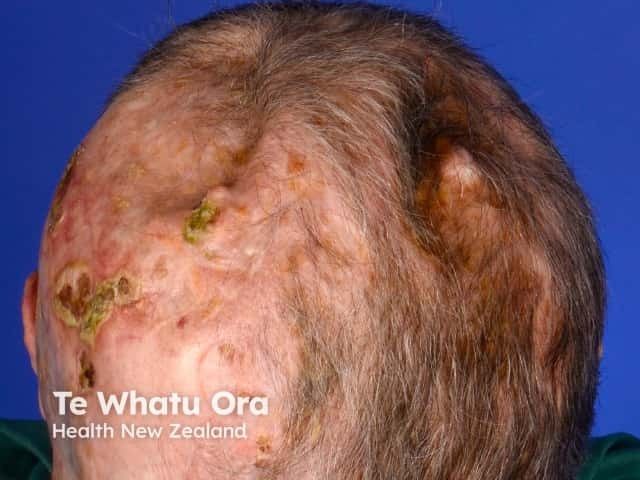
LPP affects roughly 100,000 adults in the US and currently has no FDA-approved treatments.
The ongoing phase 2b BROADEN2 study will evaluate KT-621 across 3 dose levels in 200 patients over 16 weeks, with results expected to guide phase 3 dose selection.
The combination therapy SHORE trial produced the highest response rates of the 3 studies, with EASI-75 approaching 48% in the Q4W arm.

Phase 3 data presented at AAD 2026 show once-daily oral therapy clears skin as early as week 4, offering a fast-acting option for patients.
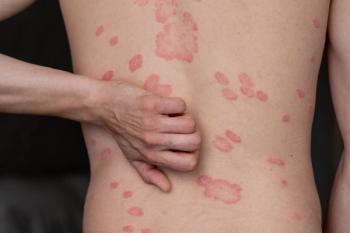
Late-breaking data shared at AAD 2026 supports oral TYK2 envudeucitinib's efficacy and safety in moderate-to-severe plaque psoriasis.
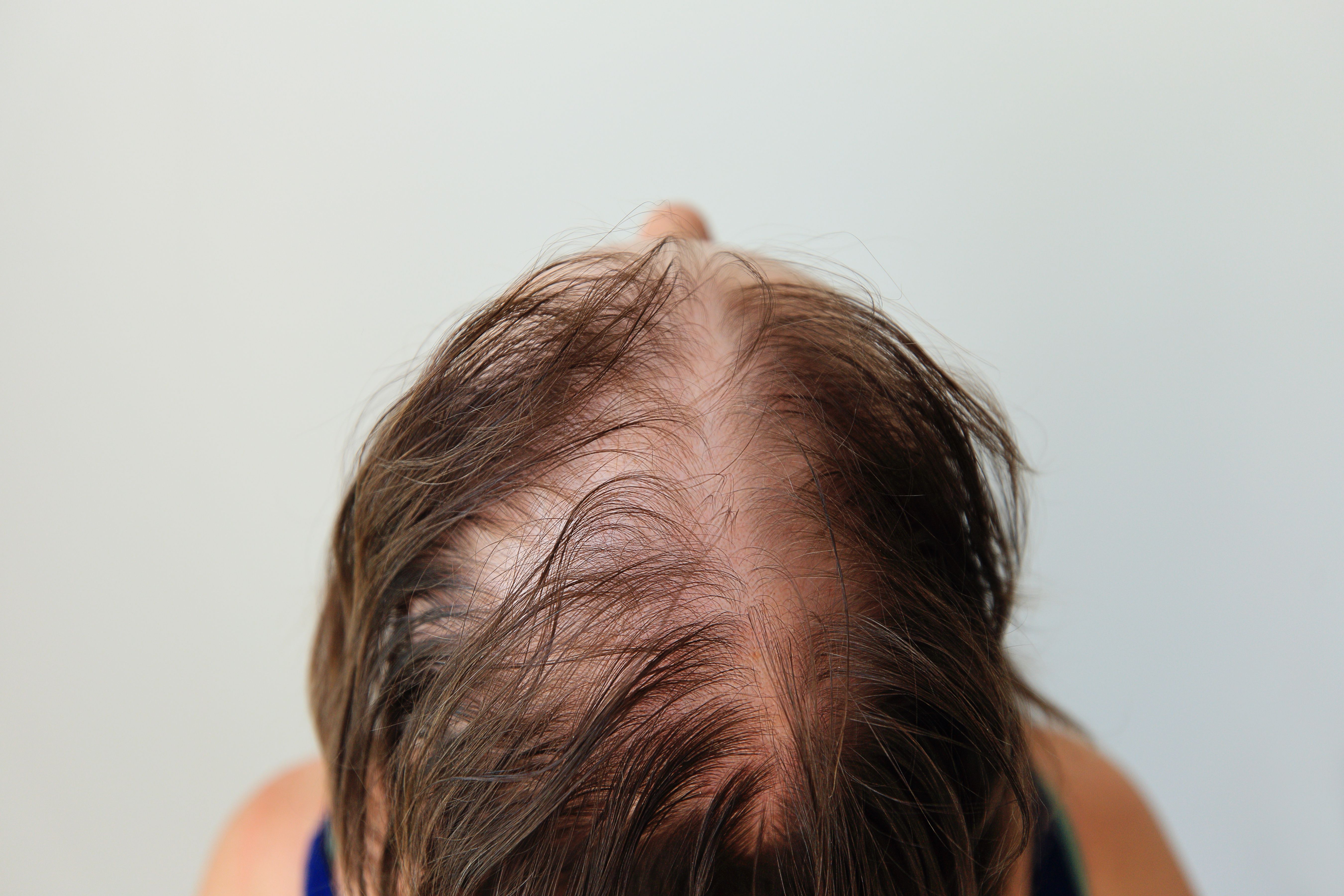
Pelage’s PP405 novel topical reawakens dormant follicle stem cells, showing signs of terminal regrowth and fueling late-stage alopecia research.

The upcoming trial will assess an injectable formulation of Xvie, distinct from its currently available topical counterpart.
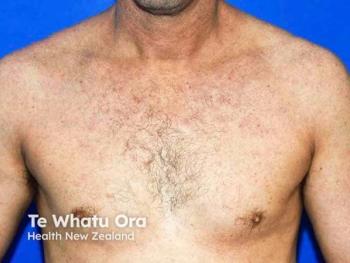
Phase 2 APEX part A data showed zumilokibart maintained EASI-75 responses in 75–85% of week 16 responders at 1 year across quarterly and biannual dosing regimens.
Only 15–20% of eligible patients currently receive systemic psoriasis treatment, and specialists believe icotrokinra's simplicity could meaningfully move that needle.
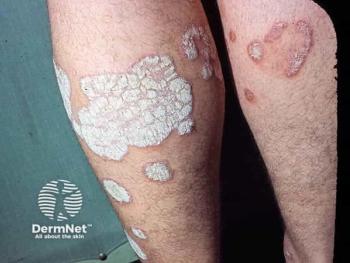
Clinical trial data showed approximately 70% of patients achieved clear or almost clear skin with icotrokinra by week 16.
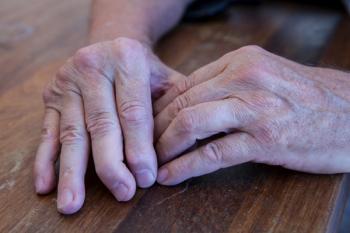
Sun Pharma's tildrakizumab nears psoriatic arthritis approval as FDA accepts a supplemental biologics license application (sBLA), spotlighting IL-23 benefits for active disease.
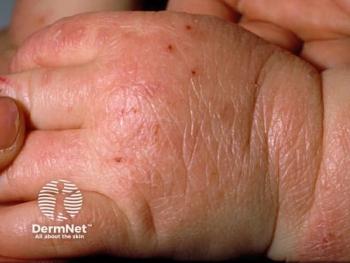
Positive phase 3 findings from ADorable-1 suggest lebrikizumab could expand biologic treatment options for younger patients with atopic dermatitis (AD).
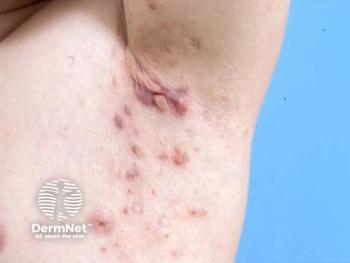
Novartis secured the approval through a pharmacokinetic extrapolation strategy rather than a full pediatric RCT, leveraging adult HS trial data and existing pediatric PK modeling from other approved indications.
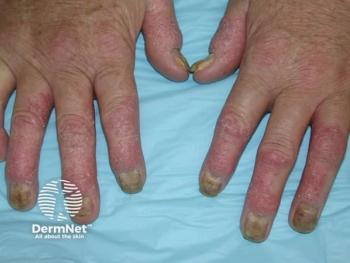
Results show that bimekizumab achieved significantly higher ACR50 response rates at week 16 than risankizumab in adults with active PsA.

FDA Fast Track boosts hopes for QRX003 lotion in Netherton Syndrome, with early trials showing rapid skin healing and itch relief.

Patients treated with deucravacitinib achieved higher ACR20 response rates at week 16 than those receiving placebo in the POETYK PsA studies.
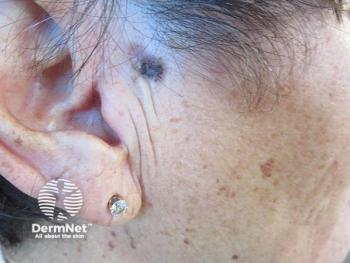
Clearance rates increased between days 29 and 57, with the highest activity observed in the 200-μg treatment group.

Rocatinlimab had met primary end points in large phase 3 trials and was previously expected to move toward regulatory submission in 2026.