Drug Watch
Latest News
Latest Videos

CME Content
More News
Almirall initiates a pivotal phase 3 trial for lebrikizumab, targeting nummular eczema and aiming to enhance treatment options for this challenging condition.

Heap discusses promising phase 3 results for zasocitinib, an oral TYK2 inhibitor, showcasing its efficacy and safety in psoriasis therapy.

Jasper Therapeutics reveals promising briquilimab data, showcasing rapid disease control and safety in chronic spontaneous urticaria and chronic inducible urticaria.
Mabwell’s investigational 9MW3811 is the first IL-11–targeting therapy to enter clinical evaluation for pathological scarring.

Explore the latest advancements in dermatology for 2026, featuring innovative therapies and treatments for skin conditions, hair loss, and more.
Topline results are expected in the first quarter of 2026.
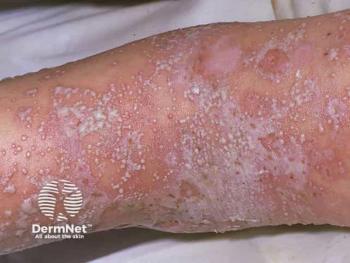
The BLA is supported by phase 3 data demonstrating rapid skin clearance and sustained disease control in patients with GPP.
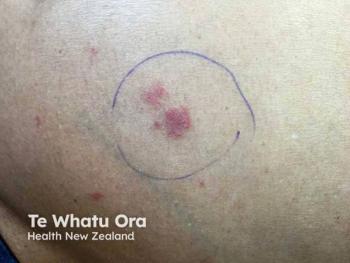
Kymera Therapeutics reveals promising phase 1b results for KT-621, an oral STAT6 degrader, showing significant effects in moderate to severe atopic dermatitis.

Abeona Initiates First Commercial Treatment of Zevaskyn for RDEB
The first treatment follows Zevaskyn’s FDA approval as the first autologous, gene-modified cellular sheet designed to address RDEB wounds.

Navigator Medicines advances NAV-240, a promising bispecific antibody for HS, targeting dual pathways for enhanced treatment efficacy.
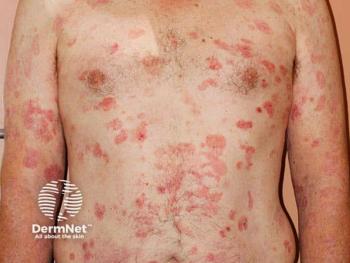
Citius Oncology’s IL-2 receptor–directed fusion protein offers rapid responses and pruritus improvement for relapsed or refractory stage I–III CTCL.

Lynk Pharmaceuticals reveals promising phase 2 trial results for LNK01004, a topical treatment showing efficacy and safety in moderate-to-severe atopic dermatitis.

New research reveals ritlecitinib's potential to reverse scarring alopecias by targeting inflammation, offering hope for hair regrowth in autoimmune disorders.

Denifanstat emerges as a groundbreaking oral therapy for acne, targeting sebum production and inflammation, promising safer, effective treatment options.

Nemolizumab shows significant efficacy in reducing pruritus and improving skin clearance in patients with prurigo nodularis, offering new therapeutic hope.
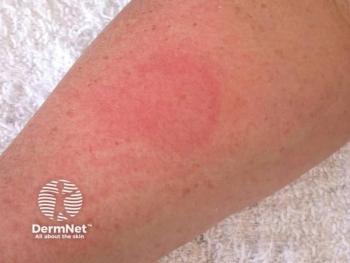
The first-in-class mast cell–selective c-Kit inhibitor ALY-301 enters clinical testing for cold urticaria in Germany.

The topline data support advancing INF904 into larger, controlled trials as a potential convenient alternative to injectable biologics.

The FDA issued 18 warning letters to websites illegally selling unapproved and misbranded botulinum toxin products, citing serious safety risks including botulism-related adverse events.
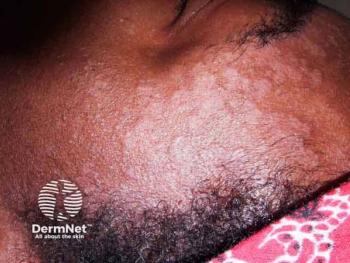
A phase 2 extension trial found roflumilast foam safe and effective for up to 52 weeks in patients with moderate to severe seborrheic dermatitis.

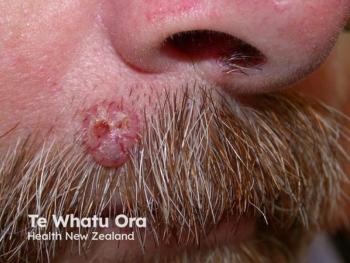
Phio Pharmaceuticals reveals promising results for PH-762, a novel siRNA therapy targeting skin cancer, showcasing effective tumor clearance and safety.

Updated FDA policies eliminate many comparative efficacy trials, reducing biosimilar development time and cost by relying more on analytical testing.

In 2 replicate phase 3 trials, once-daily upadacitinib 15 mg significantly improved total and facial repigmentation in adults and adolescents with non-segmental vitiligo, with a safety profile consistent with prior indications.

Quoin Pharmaceuticals' QRX003 granted FDA Orphan Drug Designation, advancing treatment for Netherton Syndrome, a rare dermatological disease.

Azitra reveals promising preclinical results for ATR-01, a novel treatment targeting ichthyosis vulgaris, paving the way for future clinical trials.

The CHMP opinion follows recent US FDA approval, positioning Libtayo as the first immunotherapy approved for adjuvant treatment in high-risk CSCC.