
Shawn Kwatra, MD, FAAD, reviews his late-breaking data presentation at AAD on the first original studies of a topical cream for prurigo nodularis.

Shawn Kwatra, MD, FAAD, reviews his late-breaking data presentation at AAD on the first original studies of a topical cream for prurigo nodularis.

The company showcased advancements in tirbanibulin and its early-stage antibody LAD191.

Published in JID, the study analyzes data from the FDA’s FAERS database to examine cases of neoplasms—including skin and breast cancers—potentially associated with benzoyl peroxide use.

The findings of the MVOR-1 and MVOR-2 studies are published in JAMA Dermatology, supporting the rosacea drug’s recent FDA approval.


After just 2 weeks of twice-daily use, a pediatric patient saw visually improved skin with no adverse events.

DELTA China follows the success of European and Canadian trials, reinforcing delgocitinib’s potential as a global treatment for CHE.

A PDUFA target action date has been set for October 13, 2025.

Snail slime contains bioactive compounds that promote skin healing, hydration, and regeneration.

The POETYK PSO LTE trial found deucravacitinib effective in sustaining PASI 75 and PASI 90 responses.

Alphyn’s therapy uniquely treats both the immune system and bacterial components of atopic dermatitis.

Barbieri et al evaluated product use, benzoyl peroxide concentration, days until expiration, and more.

Nektar Therapeutics’ investigational biologic targets the IL-2 receptor complex.

Many patients, especially parents of pediatric patients, exhibit steroid phobia, impacting treatment adherence.

Long-term safety data suggests that JAK inhibitors have a lower risk of cardiovascular and thromboembolic events than previously thought.

The drug, a JAK inhibitor, met all primary and secondary endpoints in a phase 3 trial for adolescent CHE.
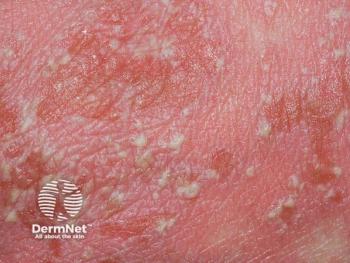
The IL-36R antagonist has demonstrated its safety and efficacy for generalized pustular psoriasis in GEMINI-1 and GEMINI-2 studies.

QRX003 mimics the LEKTI protein to strengthen skin barriers and alleviate symptoms in patients with Netherton Syndrome.

Fast-acting therapies like remibrutinib bring new hope to patients with chronic urticaria who don’t respond to antihistamines.

The new dosing option offers convenience for those managing plaque psoriasis, PsA, and hidradenitis suppurativa.

Shanna Miranti, MPAS, PA-C, discusses the Umary hyaluronic acid tablets that were recalled in fall 2024.

Mitragotri said new innovations like ionic liquid-based delivery systems can offer better treatment for immunodermatological conditions.

The innovative technology is unlocking new possibilities in dermatology by improving the solubility and permeability of drugs for topical use.

Dermatology Times is reviewing some of the most anticipated PDUFA dates of the upcoming year.

Deucravacitinib achieved ACR20 response in patients with PsA at week 16, with a safety profile consistent with previous studies.