
Abeona's pz-cel has been granted a April 29, 2025, PDUFA date.

Abeona's pz-cel has been granted a April 29, 2025, PDUFA date.

The target action review date has been pushed to March 12, 2025.

This represents the second approval of Zoryve outside the US.

Light delves into the science behind the new peer-reviewed data assessing benzene presence and formation in benzoyl peroxide products.

New peer-reviewed research published in the Journal of Investigative Dermatology examined 111 products at room temperature.

Key highlights of the presentations included data on delgocitinib for atopic hand eczema, and tralokinumab for head and neck atopic dermatitis.

Through collaboration with Nimbus Therapeutics and Schrodinger, zasocitinib’s AI-driven design maximizes its fit within the targeted enzyme.

The FDA has set a PDUFA target action date of May 22, 2025.

Bimzelx was also approved for non-radiographic axial spondyloarthritis and ankylosing spondylitis.

Ebglyss is now approved for children and adults aged 12 years and older.

The rates of discontinuation due to inefficacy or adverse events for biosimilars and originators of etanercept and adalimumab were similar, according to an analysis of a prospective registry.
The PD-1/IL-2α bispecific antibody fusion protein is intended for patients with unresectable locally advanced or metastatic melanoma.

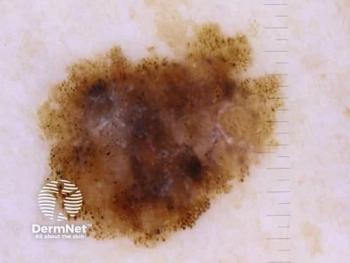
The overall reduction of tumor size in all lesions treated in part 2 was approximately 86%.

Galderma’s nemolizumab is the first approved monoclonal antibody specifically inhibiting the signaling of IL-31.

The approval is supported by positive phase 3 data for Lymphir in this indication.

Alumis' Martin Babler shared insights into the program for the TYK2 inhibitor and the company's next steps for its development.

This article represents the first time the news has been shared with the public. Turn's Founder and CEO, Bradley Burnam, shares details.

Take a deep dive into the 3 approvals for various dermatological conditions this month.

Approved down to the age of 6, the treatment is covered by 2 pharmacy benefit manager contracts to provide timely coverage.

Iltefat Hamzavi, MD, discusses the REGINA 007 phase 2 trial evaluating MK-6194 for the treatment of non-segmental vitiligo.

Along with the approval, Sun Pharma is introducing an access program to assist eligible patients in starting and adhering to the treatment.

The submission to the FDA is supported by positive results from the ARRECTOR phase 3 trial.

Patrick Burnett, MD, PhD, provides insights into the significance of Arcutis’ roflumilast cream 0.15% formulation and its impact on pediatric patients.

The planned PDUFA data was July 7, 2024.

Iain Stuart, PhD, discusses VYN202's status and potential in psoriasis and immuno-inflammatory diseases.