Conference Coverage
about 17 hours ago
Reviewing the Overarching Umbrella of Psoriatic Disease20 days ago
ASDS 2025: Top Treatments and TrendsLatest News

Journal Digest: December 17, 2025
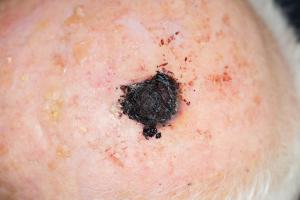
Expert Panel Unanimously Backs DecisionDx-Melanoma for Guiding Personalized Cancer Care

Reviewing the Overarching Umbrella of Psoriatic Disease
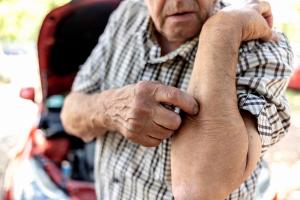
Wildfire Air Pollution Drives Surge in Clinic Visits for AD and Pruritus

Considering Biologics and Shared Decision-Making in Moderate to Severe Plaque Psoriasis: Part 2

Shorts










Podcasts
Dermatology Times Digital Edition




Continuing Medical Education
All News

In part 1 of this supplement, experts discuss current and emerging treatments for patients, considering lifestyle choices.

A bentonite and Alcea sulphurea facial mask significantly improved acne severity and patient satisfaction in a recent clinical trial.
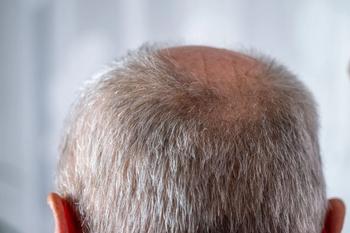
GT20029 shows promising results in treating androgenetic alopecia, enhancing hair density with minimal adverse effects in men after 12 weeks.
Topline results are expected in the first quarter of 2026.

In part 1 of this Case-Based Roundtable supplement, experts discuss biologic treatment options for psoriasis, and balancing efficacy with patient needs.
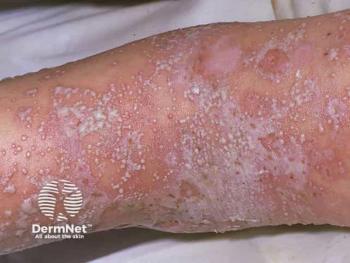
The BLA is supported by phase 3 data demonstrating rapid skin clearance and sustained disease control in patients with GPP.

Experts discuss innovative, steroid-sparing treatments for pediatric atopic dermatitis, emphasizing personalized care and proactive management strategies.

Put your dermatology expertise to the test with Dermatology Times’ interactive quiz series!

Explore the top headlines of the week, including insights on the latest clinical trials, therapeutic updates, and more.

In case you missed it, this week we had news about the first commercial treatment of Zevaskyn for RDEB, the global prevalence of HS, a new STAT6 degrader for AD, and more.

This week, we feature top articles from our sister publications on regulatory updates, clinical trial insights, and more.

John Barbieri, MD, MBA, discusses how deteriorating prior authorization processes are compromising access to essential dermatologic therapies.
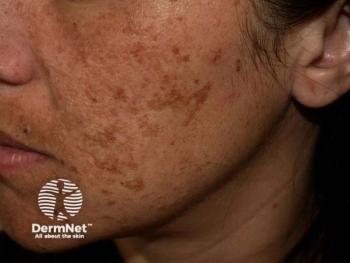
An updated review offers effective alternatives to hydroquinone for melasma treatment, including thiamidol and natural agents, with improved safety profiles.
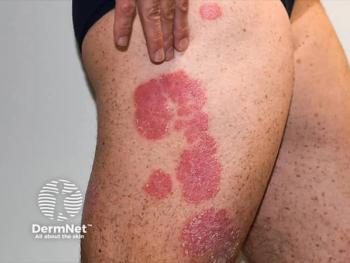
A meta-analysis of over 800,000 patients shows JAK inhibitors are generally as safe as TNF antagonists for various purposes.
































