
This week, consumer magazines highlight Botox brow lifts, the Johnson & Johnson recall of 5 aerosol sunscreen brands with benzene traces, and more.

This week, consumer magazines highlight Botox brow lifts, the Johnson & Johnson recall of 5 aerosol sunscreen brands with benzene traces, and more.

This week in aesthetics, we highlight this month’s "Meet the Aesthetic Expert" column with Morgan Wolf, DNP, FNP-BC, AGNP, Johnson & Johnson recalls 5 aerosol sunscreen brands with benzene traces, and more.

The American Academy of Facial Plastic and Reconstructive Surgery’s president Paul Carniol, MD, debunks 3 common misconceptions about facelifts.

The consumer products giant voluntarily recalled 4 Neutrogena and 1 Aveeno spray sunscreen products July 14 after internal testing found low levels of benzene, a carcinogen, in some product samples.

The consumer products giant voluntarily recalled 4 Neutrogena and 1 Aveeno spray sunscreen products July 14 after internal testing found low levels of benzene, a carcinogen, in some product samples.

A study published in Aesthetic Surgery Journal examines the efficacy of TransFORM Body Treatment with TriHex Technology over 12 weeks.

In this monthly technology review, Michael Kluska, DO, and Chad Deal, MD, discuss the EON by Dominion Aesthetic Technologies, including device specifics, cost, and ROI.

Each month, Dermatology Times® features a roundup of products trending in skin care. Click here to find out what products are trending for the month of July.
The Dermatologic Surgery journal published a study on who performs cosmetic injections at dermatology and plastic surgery practices.

In this month’s "Meet the Aesthetic Expert" column, Will Kirby, DO, FAOCD, talks with Morgan Wolf, DNP, FNP-BC, AGNP, vice president of clinical operations at LaserAway, about the increased role of allied health care professionals, the importance of continued education, and expanding diversity and inclusion in aesthetics.

With in-person conferences coming out of the shadows and into the light once again, we detail a guide to Aesthetics Biomedical’s “Perspectives. The Evolution of Aesthetics” symposium happening July 24.

The introduction of artificial intelligence into dermatology has been met with mixed emotions from the medical community. In this episode, Ade Adamson, MD, MPP, explains the benefits and detriments of the application, accuracy, and potential racial diagnostic bias.

This week, consumer magazines highlight what happens to the body while on testosterone, decoding breakouts using an ancient Chinese practice, and more.

This week in aesthetics, we announce our Summer issue, highlight female to male top surgery protocols, and more.

In this Summer issue, Paul Nassif, MD, gives his advice on how to create a successful skin care brand, Joe Niamtu, III, DMD, shares how to safeguard the practice from scams when donating to charitable organizations, and Gary Linkov, MD, shares his protocol for facial hair transplants.

Combining microwave and microablative CO2 laser treatment is both safe and effective for treating postpartum abdominal laxity.

Mark Mandell-Brown, MD, details his treatment protocol for addressing female to male breast reduction surgically and through minimally invasive procedures.

This week, consumer magazines highlight plastic surgeons accepting cryptocurrency, the difference between top surgery and a mastectomy, and more.

This week in aesthetics, we highlight the FDA approval of Restylane Contour, a guide to Aesthetics Biomedical’s upcoming “The Evolution of Aesthetics” symposium, and more.

A study compared the results of single-plane vs double-plane microfocused ultrasound with visualization (MFU-V) for upper arm skin laxity treatment.
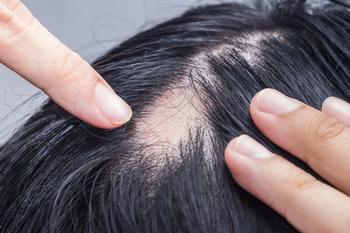
Hair regrowth was observed after starting dupilumab treatment in some pediatric patients with a history of severe and refractory alopecia areata (AA) and concomitant atopic dermatitis (AD).

With in-person conferences coming out of the shadows and into the light once again, we detail a guide to Aesthetics Biomedical’s “Perspectives. The Evolution of Aesthetics” symposium happening July 24.

Dermatologists are expanding their skill sets and exploring new techniques to enable gender-diverse patients to achieve their medical and aesthetic goals.

Galderma receives Restylane Contour FDA approval for cheek augmentation and correction of midface contour deficiencies.
Advanced Aesthetic Technologies, Inc, announces start of Algeness VL clinical trial in China seeking future market approval.