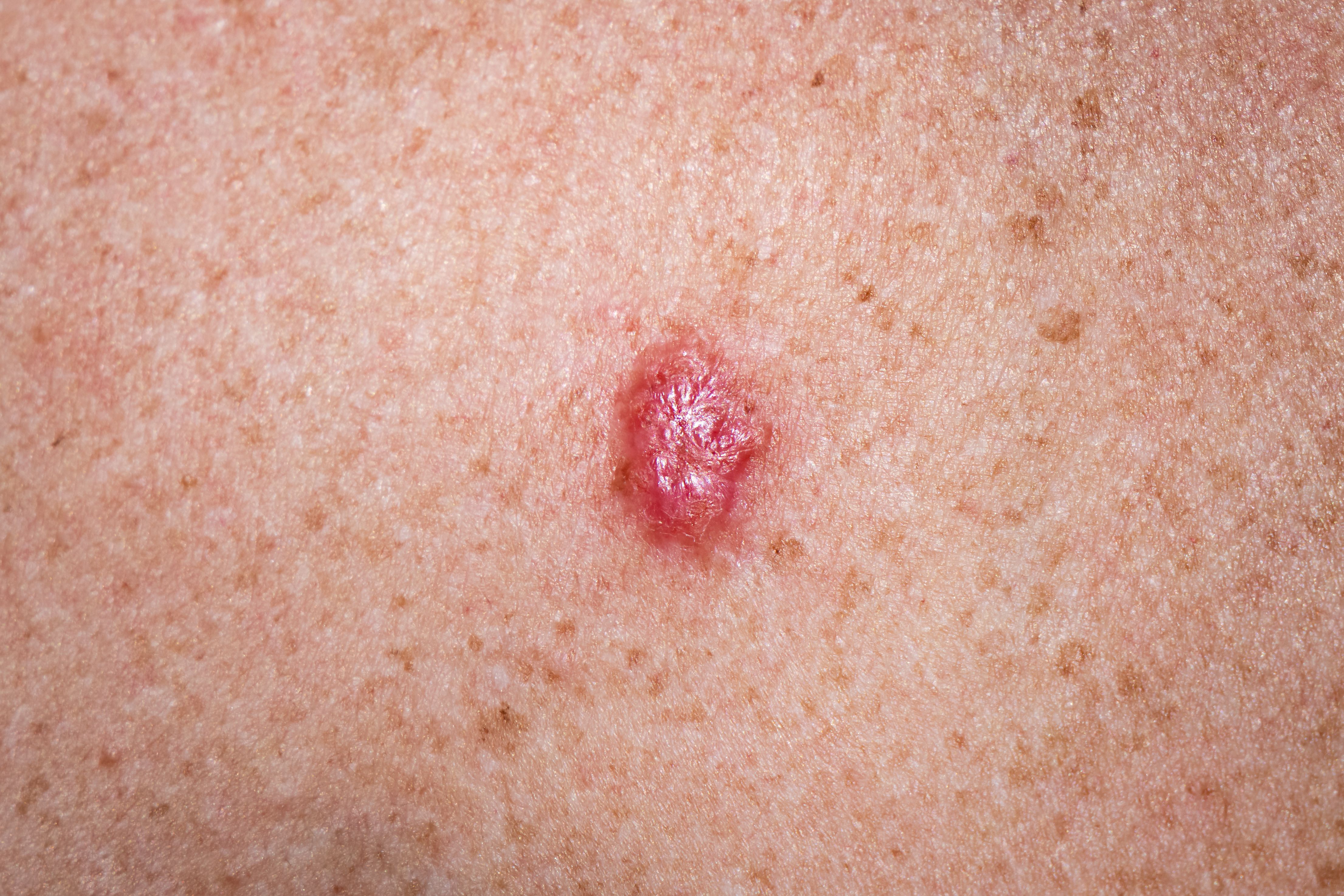
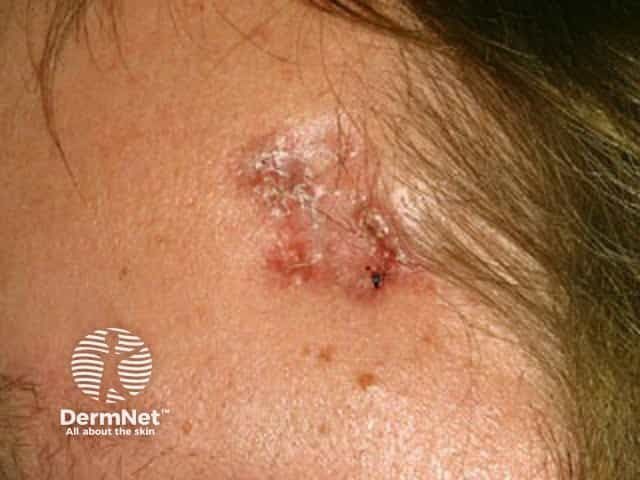
Non-Melanoma Skin Cancers
Latest News
Latest Videos

CME Content
More News

In October's cover feature, Nicole A. Negbenebor, MD, FAAD, discusses the benefits of intralesional 5-fluorouracil injection for older patients with squamous cell carcinoma who may elect to delay therapy or are not ideal candidates for surgery.

Farberg discusses data recently published in Geriatrics demonstrating low rates of recurrence.

The study, published in Dermato, showed that 92% of NMSC tumors showed measurable changes in depth of invasion from one image to the next.
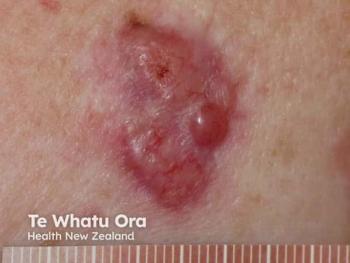
To improve overall outcomes for patients with large basal cell carcinomas, it has been beneficial to employ the use of vismodegib as a neoadjuvant and/or adjuvant treatment.

Jacob Scott, MD, president and chairman of DaRT, shares highlights of the guidelines and what he hopes clinicians can glean from them.

Margins < 1 mm also do not increase the risk of cutaneous squamous cell carcinoma recurrence risk if the galea aponeurotica is not involved.

The guidelines for BCC and SCC treatment with IGSRT detail diagnosis, staging, contraindications, protocols, and emphasize the essential role of a multidisciplinary team.

From our March cover feature: Vulnerable populations such as the prison population should have access to Mohs surgery to prevent the increased risk of morbidity and mortality from skin cancer.

New data on the devices will be available at the 2024 American Academy of Dermatology Annual Meeting.
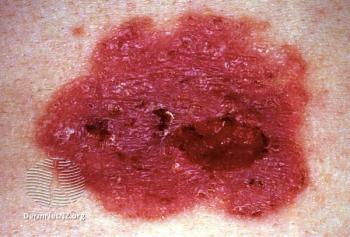
In this study, researchers treated 37 basal cell carcinoma (BCC) lesions from 30 patients, with follow-ups at 5 time points after baseline examination.

SKNJCT-003 is expected to enroll up to 60 patients.

The study evaluated the efficacy of AIV001 in superficial, nodular, and mixed BCC tumors.

One of the specific areas of focus will be non-melanoma skin cancer.

Study results were presented at the 2023 American Society for Dermatologic Surgery Annual Meeting in Chicago, Illinois.

Learn more about the in-depth topics covered in the September 2023 Frontline Forum BCC management supplement of Dermatology Times.

In the final part of this Frontline Forum series, Brent Moody, MD; Sarah Arron, MD, PhD; Justine Cohen, DO; Emily Ruiz, MD, MPH; and Todd Schlesinger, MD, review 4 real-world clinical cases of BCC and the treatments they utilized.

In part 3 of this Frontline Forum series, Brent Moody, MD; Sarah Arron, MD, PhD; Justine Cohen, DO; Emily Ruiz, MD, MPH; and Todd Schlesinger, MD, discuss non-surgical treatment options for the management of BCC, dose adjustments for systemic treatments, combining hedgehog pathway inhibitors with immunotherapy, and more.

In part 2 of this Frontline Forum series, Brent Moody, MD; Sarah Arron, MD, PhD; Justine Cohen, DO; Emily Ruiz, MD, MPH; and Todd Schlesinger, MD, discuss non-surgical treatment options for the management of BCC, dose adjustments for systemic treatments, combining hedgehog pathway inhibitors with immunotherapy, and more.

In part 1 of this Frontline Forum series, Brent Moody, MD; Sarah Arron, MD, PhD; Justine Cohen, DO; Emily Ruiz, MD, MPH; and Todd Schlesinger, MD, discuss non-surgical treatment options for the management of BCC, dose adjustments for systemic treatments, combining hedgehog pathway inhibitors with immunotherapy, and more.

Image-Guided Superficial Radiation Therapy kills cancer cells without surgery.

Collaboration between dermatologists and gynecologists can improve patient quality of life and outcomes in vSCC treatment.
Drs Sarah Arron and Nathalie Zeitouni share their insights into how they define locally advanced basal cell carcinoma and when they introduce multidisciplinary care.
Sarah Arron, MD, PhD, and Nathalie Zeitouni, MD, FAAD, discuss their approaches to managing patients with newly diagnosed basal cell carcinoma and highlight the negative consequences of a delayed diagnosis.

Click here to answer this week's poll.
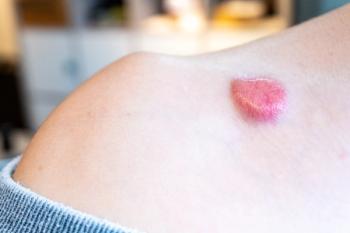
In a murine model, application of PALA in topical fashion led to reductions in tumor grades and numbers.