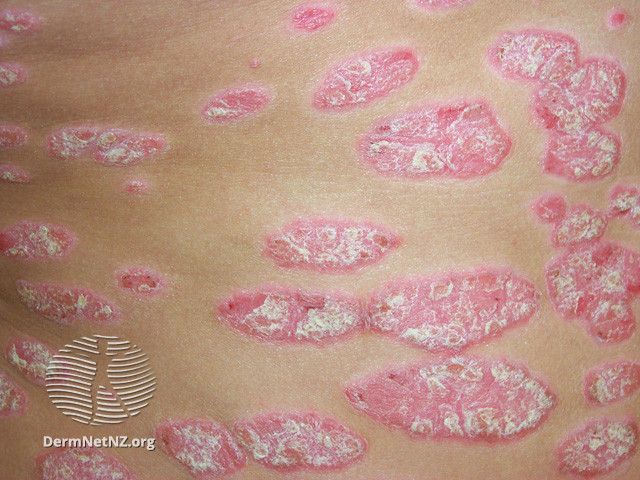
Psoriasis
Latest News
Latest Videos

CME Content
More News
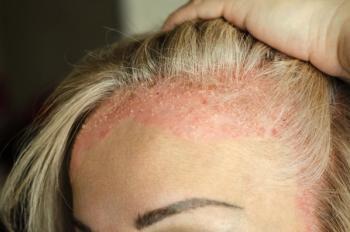
Experts in dermatology share insights on considering the location's impact in treating plaque psoriasis, focusing on challenges with scalp involvement and addressing limitations with topical therapies.

Lisa Swanson, MD, FAAD and Robert J. Casquejo, PA-C highlight the impact of plaque psoriasis on both moderate and mild patients, emphasizing the importance of addressing these concerns in patient care.

52% of adult patients achieved the treat-to-target goal of 1% BSA or less within 3 months of incorporating tapinarof cream into their biologic regimen.
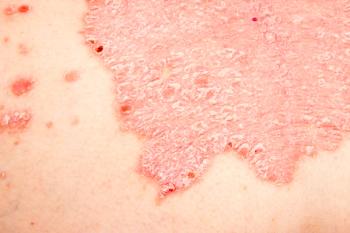
Study authors hypothesize that a monoclonal blockade of pathogenic T cells may induce expansion of regulatory immune cells subsets or expression of cytokines involved in skin homeostasis.

Investigators explored the genetic relationship between psoriatic disease and serum lipid levels.
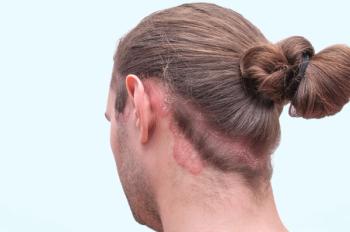
Approximately 96.2% of patients had achieved PASI75 by week 12 of the study.
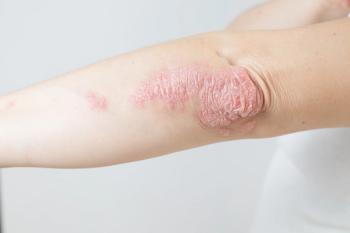
Researchers explored the potential causal relationship between pure hypercholesterolemia, a recognized risk factor for cardiovascular disease, and psoriasis.

The US license period will start on February 22, 2025.
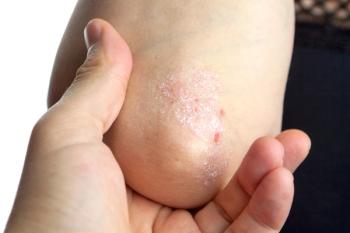
Depression, functional impairment, and worsened quality of life are related in patients with psoriatic disease, according to a recent study.
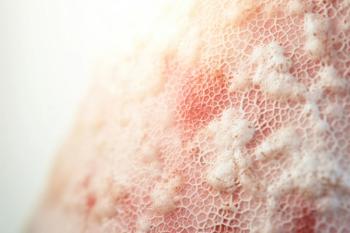
Because of the chronic inflammatory state of psoriasis and the immunosuppression produced by some systemic medications, patients with psoriasis may have an increased risk of lymphoma and nonmelanoma skin cancer.

Label warnings may affect the initiation of new treatment and real-world switching patterns for psoriasis patients starting treatment with risankizumab.

Bimekizumab is the first dual IL-17 A/F inhibitor to treat moderate-to-severe plaque psoriasis. It launches with a list price of $7,200 per syringe.

In addition to improvements in work productivity, tildrakizumab also demonstrated significant efficacy and safety.

Investigators noted that patients with mild psoriasis experience a less favorable status when compared to patients with moderate psoriasis.

Psoriasis Patients With Pruritis, Sleep Impairments Demonstrate Lower Quality of Life in All Domains
PASI scores were significantly correlated with sleep quality, duration, and disturbances.

Authors of the case report that little is known about the associations between psoriasis and renal function.

The approved indication makes Wezlana the first approved interchangeable biosimilar to Stelara.

Leah Howard, JD, of the National Psoriasis Foundation, discusses the importance of advocacy in psoriasis, the role of mental health in chronic skin conditions, and the role of support organizations in advocating for awareness and health equity.
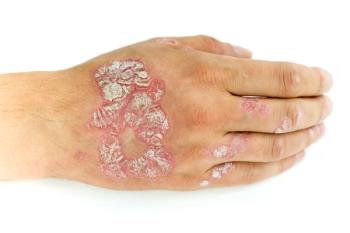
Tapinarof Cream 1% Led to Rapid Improvements in Itch for Adults With Mild to Severe Plaque Psoriasis
Once daily application of tapinarof cream 1% rapidly improved itch in adult patients with varying plaque psoriasis diseases severity, according to a poster presented at SDPA’s fall conference.
Douglas DiRuggiero, DMSc, PA-C, presented a comprehensive session at the SDPA fall conference on treatment selection for patients with psoriasis.

Black and male patients exhibited a higher prevalence of palmoplantar psoriasis when compared with White patients.

82.8% of adult patients with iPsO achieved an IGA score of 0 or 1 indicative of complete or almost complete lesion clearance.

Topline results from the phase 3b trial revealed significant improvements in patients with skin of color whose psoriasis was treated with guselkumab.

Bimekizumab is now the first and only approved IL-17A and IL-17F inhibitor for this indication.

At week 8, 65.3% of roflumilast-treated patients achieved a clinically significant reduction in itch.