
Acne
Latest News
Latest Videos

CME Content
More News
Dr Joshua Zeichner reviews the mechanism of action of Isotretinoin and highlights utilization requirements in patients prescribed this treatment.

Joshua Zeichner, MD, shares his insight into some causes of acne and the utility of clascoterone cream 1% combat this condition.

An expert in acne management provide his opinion on the differences between generic vs brand acne treatments and shares his thoughts on the utilization of available isotretinoin formulations.

Aaron Farberg, M.D discusses some common acne causes, highlighting the role of stress, diet, and androgen receptors for this condition.

Dr Aaron Farberg shares his recommendations for discussing acne treatment options with patients and provides insight on setting and managing patients’ goals and expectations.

Dr Leon Kircik, comments on the impact the multiple formulations of isotretinoin had on acne care; and highlights the efficacy differences between generic vs brand formulations.

Leon Kircik, M.D., discusses the indication of isotretinoin for use in patients suffering from acne, highlighting the difference between high and low dose administration needs.

An expert in acne management shares her insight into the patient characteristics a provider needs to consider when assessing the risk/benefit of a therapeutic agent for the management of acne.

Dr Zoe Diana Draelos discusses the role of ancillary skin care products in the management of acne and stresses the importance of setting patient expectations.

Zoe Diana Draelos, MD comments on the importance of tailoring acne treatments based on disease severity and the ability to target key acne pathogenesis factors.

Truncal acne, or back acne, is a common skin condition of the pilosebaceous unit, with a global lifetime prevalence of 70% to 85%.
Hilary Baldwin, M.D., shares her hopes for the future of acne treatment and provides her practice pearls from a panel discussion surrounding the management of patients suffering from acne.

An expert in acne management shares her insight on the current needs and the role of consensus guidelines in the management of patients suffering from acne.

Dr Hilary Baldwin discusses her treatment considerations and approach in the management of patients with mild to moderate acne, highlighting the benefit of combination therapy.

Hilary Baldwin, M.D., comments on the impact of the recent approval of clascoterone cream 1% in the topical acne armamentarium of patients suffering from acne.

CBD therapy has had positive outcomes in other inflammatory conditions.
Newly approved laser treatments can be an effective option for the treatment of this common skin disorder.

Accure’s system is the first 1726nm-based laser device with both FDA and CE Mark clearances.
Numerous adverse effects are associated with standard-dose isotretinoin.
Patient-reported outcome measures ensure patients' experiences are documented during new treatments.

Neal D. Bhatia, MD, discusses the nuances of photodynamic therapy in a session at Fall Clinical in Las Vegas, Nevada, October 20-23.

Understanding the main causes and staying current on recommendations and treatments will help reduce patient rates of the condition.
New studies explore how stress plays a large role in triggering acne.
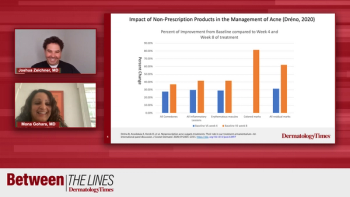
Drs Joshua Zeichner, MD, and Mona Gohara, MD, provide take-home messages to providers treating patients with acne.

Expert dermatologists discuss different types of moisturizers and how to properly select a moisturizer.























