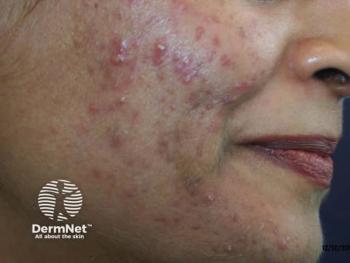
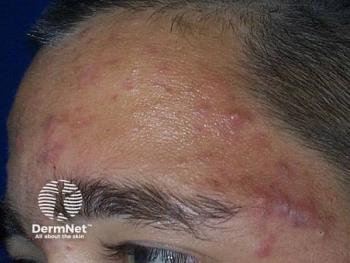
New Pediatric Perspectives for Acne
Experts at Maui Derm Hawaii offer the latest findings and research in treating acne vulgaris in adolescents.
In a shared session at the 2023 Maui Derm Hawaii conference, Lawrence Eichenfield, MD, Professor of Dermatology and Pediatrics, and vice chair at Rady Children’s Hospital in San Diego, California, examined commonly used treatments and new therapies for the treatment of acne vulgaris in children and adolescents.
He reminded attendees that acne vulgaris is classified based on patient age, lesion morphology (ie, comedonal, inflammatory, mixed, nodulocystic), distribution (ie, location on face, trunk, or both), and severity (ie, extent, presence or absence of scarring, postinflammatory erythema, or hyperpigmentation).1
Eichenfield’s session discussed several commonly used treatments for acne in pediatrics, all of which have been approved for patients ages 9 and older; they all are typically well-tolerated with no significant differences in responses between age groups.
- Trifarotene 0.005% cream is a retinoid indicated for the topical treatment of acne vulgaris. It has been studied in cases of facial acne, as well as chest and back acne.
- Tazarotene 0.045% lotion was developed using polymeric emulsion technology to provide a well-tolerated topical retinoid.
- Minocycline is a tetracycline antibiotic that is a highly effective oral treatment for acne. It has also been approved for non-nodular moderate-to-severe acne for children ages 9 and older. The drug uses molecule stabilizing technology; investigators have not found evidence of phototoxicity, photoallergy, or skin sensitization.
- Clascoterone cream 1% is indicated for acne in patients 12 years and older. According to the US Food and Drug Administration, more patients achieved a reduction in acne as well as clear or almost clear skin after 12 weeks of treatment in comparison to those who received a placebo. This drug is rapidly metabolized into the skin with no anti-androgenic activity. It has been shown to inhibit sebum production, reduce inflammatory cytokines, and inhibit inflammatory pathways.
Eichenfield also spoke of sarecycline (Seysara), a tetracycline-class medication. It is labeled for the treatment of inflammatory lesions of non-nodular moderate to severe acne vulgaris in patients nine years and older.
Eichenfield said the use of laser therapy also should be considered for the treatment of acne in adolescents. He listed several currently in use, including
- Blue Light
- Pulsed Dye Laser ( PDL)
- Nd: YAG (Long-pulsed; Q-switched; Combination)
Erbium Laser Resurfacing (ERB)
- Intense Pulsed Light (IPL)
- Photodynamic Therapy
- Solid-State Fractional 589/1319 nm laser
- 1726-nm laser systems, an energy based-device for the treatment of mild, moderate, and severe acne.
In the presentation, Eichenfield updated attendees with new data from a recent 1726-nm laser systems study. After 3 30-minute sessions, 90% of patients had visible improvement in their acne at 6 months. New 12 month data shows improvement increased to 92%, with 75% of patients clear or almost clear at 12 months.
Eichenfield remains excited about new technologies and treatments in the field, and told attendees there is always something new clinicians can learn about treating acne in youths. All cases of acne should be treated individually, based on genetics and severity of the patient’s acne vulgaris, he said.
Reference
1. Eichenfield, Lawrence. What’s New in Acne: Pediatric Perspectives. Presented at Maui Derm Hawaii 2023 Conference; January 23-27, 2023. Maui, Hawaii.