
The goal of Arron et al’s study was to determine if the 40-GEP test could identify high-risk cSCC patients who would benefit from ART in controlling metastatic disease progression.

The goal of Arron et al’s study was to determine if the 40-GEP test could identify high-risk cSCC patients who would benefit from ART in controlling metastatic disease progression.

Farberg is looking forward to another year of top-notch presentations at Fall Clinical 2023.

The advancement is based on previous observations of preliminary clinical efficacy. There are currently no FDA-approved therapies for this patient population.

Approval is based on data from Bristol Myers Squibb’s CheckMate – 76K trial.

Aaron Farberg, MD, and Shanna Miranti, MPAS, PA-C, discuss the upcoming approvals of nivolumab and IDP-126.
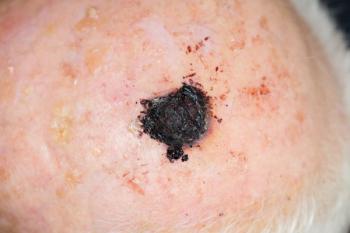
Greater treatment costs are associated with more advanced stages of melanoma, particularly due to increasing prices of treatments.

Click here to answer this week's skin cancer poll.
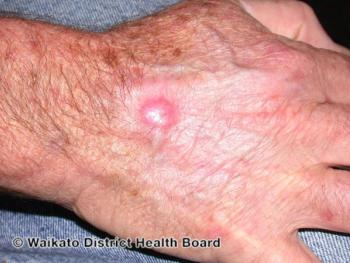
Surgical removal techniques including Mohs surgery and excisional surgery are preferred treatment approach when addressing BCCs, particularly on the hands.

Dermatologist Laura Ferris, MD, PhD, discusses skin-related toxicities as a result of immunotherapies for the treatment and management of cancers.
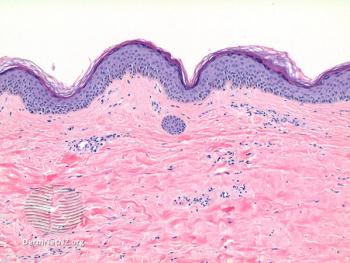
Many studies have investigated the associations between lichen sclerosus and vulvar and penile SCCs, as well as genital melanomas.

In this week’s Pointers With Portela, the 208SkinDoc explains why individuals with certain careers have an increased risk of developing skin cancer.

Learn more about the in-depth topics covered in the September 2023 Frontline Forum BCC management supplement of Dermatology Times.

In the final part of this Frontline Forum series, Brent Moody, MD; Sarah Arron, MD, PhD; Justine Cohen, DO; Emily Ruiz, MD, MPH; and Todd Schlesinger, MD, review 4 real-world clinical cases of BCC and the treatments they utilized.

In part 3 of this Frontline Forum series, Brent Moody, MD; Sarah Arron, MD, PhD; Justine Cohen, DO; Emily Ruiz, MD, MPH; and Todd Schlesinger, MD, discuss non-surgical treatment options for the management of BCC, dose adjustments for systemic treatments, combining hedgehog pathway inhibitors with immunotherapy, and more.

Frequent fliers, including pilots and cabin crew, are exposed to greater levels of UV and cosmic ionizing radiation than the general population, leading to higher incidence of skin cancers.

In part 2 of this Frontline Forum series, Brent Moody, MD; Sarah Arron, MD, PhD; Justine Cohen, DO; Emily Ruiz, MD, MPH; and Todd Schlesinger, MD, discuss non-surgical treatment options for the management of BCC, dose adjustments for systemic treatments, combining hedgehog pathway inhibitors with immunotherapy, and more.

In part 1 of this Frontline Forum series, Brent Moody, MD; Sarah Arron, MD, PhD; Justine Cohen, DO; Emily Ruiz, MD, MPH; and Todd Schlesinger, MD, discuss non-surgical treatment options for the management of BCC, dose adjustments for systemic treatments, combining hedgehog pathway inhibitors with immunotherapy, and more.
Rescue workers had a higher rate of malignant melanoma between 2005 and 2015 compared the general population of New York State.
Second-line end points of the TRICOTEL trial assessing atezolizumab plus vemurafenib and cobimetinib in melanoma with central nervous system metastases appear consistent with primary efficacy end points.
A coprimary end point in the phase 3 LEAP-010 study will not be met.

Click here to answer this week's skin cancer poll.
Melanoma cases were almost 10 times higher in high Human Development Index countries than in low.

In this week’s Pointers With Portela, the 208SkinDoc takes an in-depth look at Merkel cell carcinoma after Jimmy Buffett passed away from a 4-year battle with the rare form of skin cancer.

Endocrine toxic effects were more likely than nonendocrine to become chronic and continue at last follow-up.

Risk of recurrence or death was reduced by 58% compared to placebo.