- Dermatology Times, October 2019 (Vol. 40, No. 10)
- Volume 40
- Issue 10
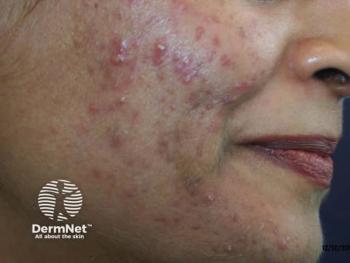
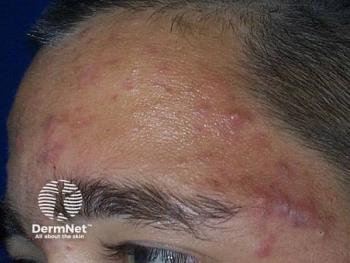
New topical retinoid for acne achieves efficacy endpoints
Phase 3 studies suggest new topical retinoid effectively treats facial and truncal acne.
Two phase three studies suggest once-daily trifarotene 50 µg/g cream effectively and safely treats moderate facial and truncal acne.
Researchers reported on both 12-week double-blind, randomized, vehicle-controlled studies on people ages 9 years and older in a paper published June 2019 in the Journal of the American Academy of Dermatology (JAAD).1 The studies, called PERFECT 1 and PERFECT 2, included 200 sites in the United States, Canada, Europe and Russia and enrolled 2,420 patients with moderate facial and truncal acne who were treated with either trifarotene or vehicle.
Trifarotene (Galderma) is a retinoid receptor agonist that selectively targets retinoic acid receptor gamma (RAR-γ), which distinguishes it from other topical retinoids that target both retinoic acid receptor beta (RARβ) and RARγ, according to the paper.
“Trifarotene is pharmacokinetically stable in keratinocytes but is rapidly metabolized in hepatic microsomes, predicting a favorable safety profile; in addition, it has comedolytic, anti-inflammatory and anti-pigmenting properties,” the authors write.
The study design stands out because, while the pathophysiology and presentation of facial and truncal acne are thought to be similar, acne treatment options have not been vigorously studied on truncal acne, the authors write.
The primary efficacy endpoints of the studies were Investigator’s Global Assessment (IGA) success rate on the face, of clear/almost clear and at least a two-grade improvement from baseline at week 12, and absolute change from baseline in facial inflammatory and non-inflammatory lesion counts from baseline to week 12. Secondary efficacy endpoints were Physician Global Assessment (PGA) success rate on the trunk, with clear/almost clear and at least a two-grade improvement from baseline, at week 12, and absolute change in truncal inflammatory and non-inflammatory lesion counts from baseline to week 12, according to a Galderma press release.
Researchers in both studies reported that trifarotene was superior to vehicle in all primary and secondary efficacy assessments. It was superior to vehicle in reducing inflammatory and noninflammatory lesion counts on the face and trunk. Trifarotene 50 µg/g cream worked quickly, according to the study, significantly reducing inflammatory and noninflammatory lesion counts in as little as one week of treatment on the face and two weeks of treatment on the trunk.
Treatment with the investigational cream was relatively tolerable, manageable and consistent with topical retinoid dermatitis. Adverse events led to 1.9% of patients in the PERRFECT 1 trial and 1.2% of those in the PERFECT 2 trial to discontinue trifarotene treatment. No patients in the vehicle groups discontinued due to adverse events.
“Trifarotene represents a novel topical retinoid with demonstrated efficacy in patients with moderate facial or truncal acne,” says Diane Thiboutot, M.D., an investigator in the phase 3 clinical trials for trifarotene and professor of dermatology at Penn State College of Medicine, Hershey, Pa.
Dr. Thiboutot says she thinks trifarotene’s role in acne treatment would be for patients with moderate acne comprised of either comedones with papules, pustules or both.
It’s not so much that trifarotene would replace current acne treatments because there’s so much variability to patient response from treatment, she says.
“Rather I view [trifarotene] as an effective option for patients that haven’t responded to other topical retinoids. I also view it as an alternative to topical antibiotics and in some cases an alternative to oral antibiotics,” Dr. Thiboutot says.
Like with other retinoids, clinicians prescribing trifarotene should educate patients about typical topical retinoid side effects. This includes educating patients about the proper amount of medication to use, how to apply it and how to incorporate the use of moisturizers into their skincare regimens, she says.
Trifarotene is in clinical development for the treatment of moderate facial and truncal acne, including large body surface areas, according to a Galderma press release.
Disclosures:
Disclosures: Nestle Skin Health Care-Galderma funded the phase 3 studies. Galderma employees were among the studies’ authors. Dr. Thiboutot was an investigator in the Phase 3 clinical trials for trifarotene at Penn State and attended one advisory meeting for trifarotene. She also served as an investigator for Botanix, Foamix and Cassiopea.
References:
Reference:
1 Tan J, Thiboutot D, Popp G, et al. Randomized phase 3 evaluation of trifarotene 50 μg/g cream treatment of moderate facial and truncal acne. J Am Acad Dermatol. 2019;80(6):1691-1699.
Articles in this issue
over 6 years ago
Non-melanoma skin cancer increases in the elderlyover 6 years ago
Looming medical billing and coding changes in healthcare policyover 6 years ago
7 tips to get the most from your social media postsover 6 years ago
IL-17 inhibitors may fill treatment gap in rosaceaover 6 years ago
Head and neck melanoma characteristics