
The Food and Drug Administration(FDA) approved in December a new drug application for Tolsura (SUBA-itraconazole), a new formulation of itraconazole for the treatment of systemic fungal infections in adults.

The Food and Drug Administration(FDA) approved in December a new drug application for Tolsura (SUBA-itraconazole), a new formulation of itraconazole for the treatment of systemic fungal infections in adults.

Fillers and skin tightening technologies provide safe, synergistic results.

With the recent FDA approval, Jeuveau execs believe they’ve got a brand that could neutralize the existing monopoly.

The FDA has approved STP705, an siRNA (small interfering RNA) therapeutic for in situ Squamous Cell Carcinoma Nonmelanoma Skin Cancer (NMSC), to proceed with phase two clinical trials.

Vyome Therapeutics Inc. announced in January that it secured $22 million in financing to, in part, support the development of its lead molecule, VB 1953, through phase two clinical trials for moderate to severe acne.

Industry relationships are essential for innovation in dermatology. In this month's column, Steve Xu, M.D., and Michael L. Sierra, Ph.D., provide their insight on collaborating with industry.

According to recent research, smokers are more likely than nonsmokers to have complications after undergoing cosmetic surgery on the body, versus the face or breasts.

A recent study suggests that patients suffering from psoriasis and psoriatic arthritis may have an increased risk of developing autoimmune thyroid disease.

Doctors should be vigilant about recognizing the signs and symptoms of patients who may develop ILD in order to prevent catastrophic complications, such as respiratory failure.

Outpatient dermatology services can be available to any patient. However, according to recent research, that doesn’t mean all patients access or take advantage of this clinical care in the same way.

Robot dermatologists aren’t yet seeing patients for routine care, but artificial intelligence (AI) and other technology tools are growing in popularity throughout the industry. Learn more about how these advances in technology are impacting practices.

Artificial intelligence (AI) tools are steadily growing in popularity throughout medicine, particularly in dermatology. But does AI pose a threat to dermatologists? Find out in this article.

In this article, Katja Reuter, Ph.D. critiques a study by Katz and colleagues on the effectiveness of using digital platforms to recruit for an atopic dermatitis study.

Apps aren’t just for mindless time-killing. A slew of apps are available to help you move more, eat better, and sleep deeper.

Do you know the warning signs of life threatening purpura? Dr. Roderick Hay outlines what you need to know about potentially life-threatening conditions involving purpura.

Digital recruitment for clinical studies may be more effective than traditional methods, according to a new study.

What exactly is digital health? Dr. Steve Xu tackles this question in this month's Innovation column.

Sandoz, a division of Novartis, announced that the U.S. Food and Drug Administration approved its biosimilar, adalimumabadaz (Hyrimoz, Novartis AG).
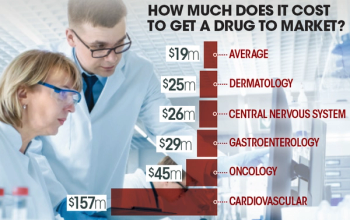
The cost of clinical trials to get a dermatology drug to market may cost as much as $25 million, which is slightly above a median of $19 million, according to the latest research.
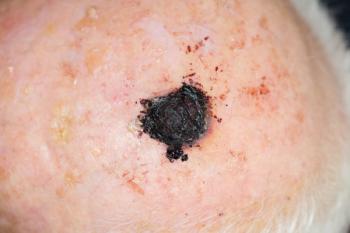
What is a life worth? Does it come down to dollars and cents? A recent study examines these questions in light of current and projected costs of melanoma treatment.
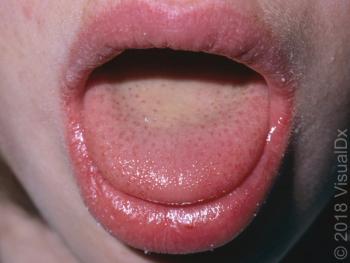
Image IQ: An 8-year-old girl with a sore throat, deep red tongue, fever and a petechial rash. What’s your diagnosis?

Before 2018 comes to a close, we wanted to say thank you to our readers from everyone here at Dermatology Times. As a thank you, we have compiled a list of the topics and issues you found most compelling throughout the year. Enjoy this slideshow - and see you in 2019!


While neuropathic pain is a focus of research and drug development, the same cannot be said for neuropathic itch.

European Union regulation on data protecion has put pressure on U.S. legislators and regulators to heighten protections within our own borders, experts say. The ramifications for healthcare organizations, both abroad and in the United States, are significant. Here’s what you need to know.

Researchers explore the association between drug price and market competition among the most commonly prescribed topical generic medications in dermatology. Here's what they found.

The TNF alpha inhibitor certolizumab pegol (Cimzia, UCB) has achieved the highest response rates seen in phase three trials of self-injectable biologics for psoriasis.

Biologic tildrakizumab-asmn approved with suffix as differentiator from future biosimilar versions.

The use of cloudbased platforms to send, receive and store patient data in teledermatology adds security risks.

A 13-year-old girl presented with a history of atopic dermatitis and alopecia totalis. Nine months after starting dupilumab, the patient regrew hair on 60 percent of her scalp.