Aesthetics
Latest News
Latest Videos

CME Content
More News

Sachin Shridharani, MD, FACS, discusses the READY-1 and READY-2 clinical trials and positive support for Relfydess.
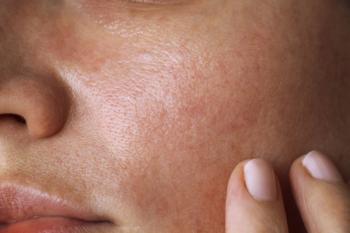
ND-ZnO technology also demonstrated substantial skin permeation and active ingredient delivery.

A group of international experts focused on the technology’s potential to standardize and enhance patient assessments.
Explore solutions for facial volume loss, skin laxity, and in some cases—skin excess.
Researchers noted that many dermatologists face mild, non-life-threatening complications during procedures, yet must be prepared for emergencies.

Zoe Diana Draelos, MD, examines common questions and controversies regarding physiologic differences in skin between White, African American, and Hispanic individuals.
Walter Liszewski, MD, shares insights into discrepancies between permanent makeup and decorative tattoos and which patients may benefit from permanent makeup.

The recent news of counterfeit Botox resulted in patients with blurred vision, difficulty swallowing, shortness of breath, and additional symptoms.
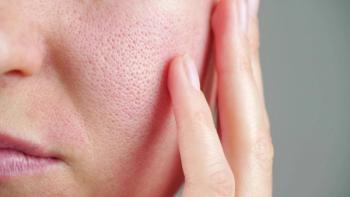
In this month's Cosmetic Conundrums column, Zoe Diana Draelos, MD, examines the most popular active ingredients in redness-reducing formulations.

Robin Siperstein, MD, FAAD, shares practical tips in cosmetic dermatology at the 2024 SDPA Annual Summer Dermatology Meeting.

In this month’s Cosmetic Conundrums column, explore what dermatology clinicians need to know about false eyelashes to help prevent infections or contact dermatitis.

These results are indicative of the need for dermatologists to discuss social media misinformation and low quality information with their patients.

This week’s collection of the latest dermatologic studies includes artificial intelligence in cutaneous lesions, dupilumab-induced acanthosis nigricans, aesthetic considerations for treating lesbian, gay, and bisexual patients, and monotherapies for male androgenetic alopecia.

Renata Block, MMS, PA-C, shares her favorite topics from the aesthetic conference held last month.

While 84% of commercials targeted female consumers and 77% utilized celebrity influence, less than half mentioned specific ingredients in skin lightening products.

A recent survey covered topics including skin-aging prevention, care-seeking behavior, trust in professionals, and more.

The FDA has released guidelines for clinicians and tips to identify the distinguishing features of counterfeit products.
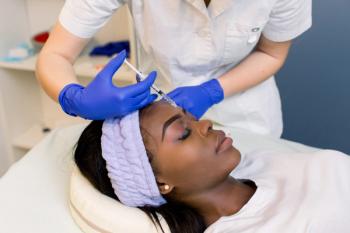
In a literature review, researchers found data supporting the efficacy and safety of fillers in patients with skin of color, particularly for populations where is there is limited data.

In this study, topical 0.1% retinol significantly improved crow’s feet, facial wrinkles, uneven skin tone, brown spots, and overall photoaging.

Shah discusses the science behind his products, as well as the challenges he didn’t anticipate when launching Remedy.

Moving the Needle on Ethics includes articles and interviews featuring experts in aesthetics.

Amy B. Lewis, MD, explores the evolution of aesthetic dermatology, delving into injection sophistication, devices, and top procedures.

In her March edition of Cosmetic Conundrums, Zoe Diana Draelos, MD, explores the role of exosomes in aesthetic dermatology, changes in the skin, and more.

Founder Dina El-Sherif, MBA, gave an in-depth overview of the at-home facial rejuvenation device and its 3-step process at AAD 2024.

The poster, which shares results form consensus research on using LODM for hair loss, was awarded 4th place at the 2024 AAD Annual Meeting.