
For the first time, there are trials looking at therapies that actually heal epidermolysis bullosa (EB) wounds, offering hope for patients suffering from the rare genetic disorder.

For the first time, there are trials looking at therapies that actually heal epidermolysis bullosa (EB) wounds, offering hope for patients suffering from the rare genetic disorder.

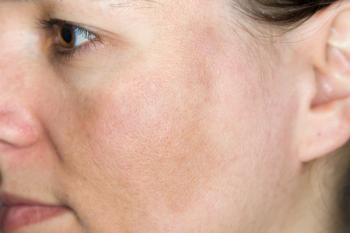
Recent data sheds light on the pathogenesis of rosacea and outlines treatment options to better manage patients.
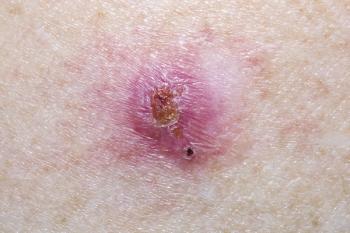
Researchers investigated the long-term efficacy of low-dose vismodegib in locally advanced basal cell carcinoma patients who achieved complete remission.

Vince Bertucci, M.D., offers tips for building trust, addressing concerns during a cosmetic patient consultation.
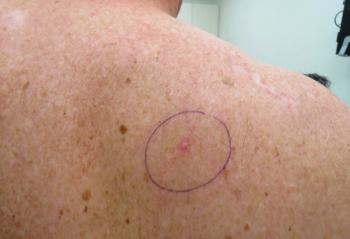
For nonaggressive basal cell carcinomas, laser therapy could be a practical treatment and management solution, according to the authors of a recent review.
Jonathan H. Zippin, M.D., Ph.D., explains what existing drugs and investigational compounds that inhibit the JAK-STAT pathway are being investigated as treatment for atopic dermatitis, alopecia areata, vitiligo and psoriasis.
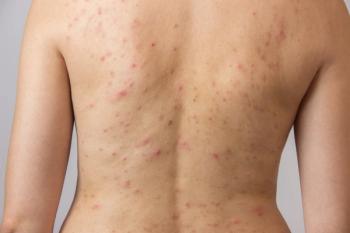
The topical combination of tretinoin 0.05% lotion and azelaic acid 15% foam improved truncal acne and may be an alternative to oral antibiotic treatment, according to recent case studies.

Efforts are underway to identify new therapeutic methods that can offer improved outcomes for patients with hidradenitis suppurativa.
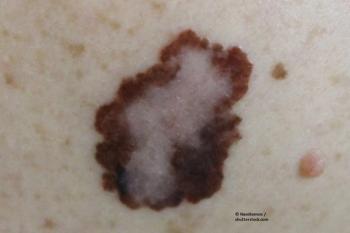
Recognition of patients with hereditary melanoma is important not only because these individuals require careful surveillance for future primary melanomas, but also because they may be predisposed to multiple other types of malignancies because of their genetic mutation, says Hensin Tsao, M.D., Ph.D.
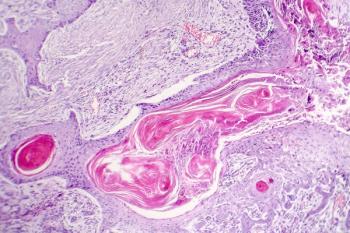
A new energy-based device delivering ultrafast pulses of high amplitude electrical energy to nonthermally preferentially clear targeted cell rich lesions is proving to be a very useful treatment modality for a number of benign dermatologic lesions.

A third-year resident in the department of dermatology at the University of California, Irvine has kicked off a passion project turning discarded sterilization wrap into face masks with a high filter efficiency. She hopes these will fill a need as well as reserve N95 masks and surgical masks for those procedures that they are designated.
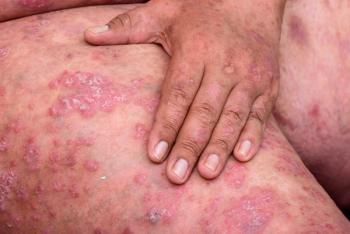
Amgen releases positive phase 3 results evaluating U.S. FDA-approved apremilast (Otezla) for the treatment of mild-to-moderate plaque psoriasis in adults.

LEO Pharma announces azelaic acid foam 15% (Finacea) is now available on the market to treat inflammatory papules and pustules of mild-to-moderate rosacea.

Brodalumab, guselkumab, ixekizumab and risankizumab stood out among 15 biologic and oral medications as having the highest short- and long-term response rates for the treatment of moderate-to-severe plaque psoriasis, according to a recent meta-analysis.
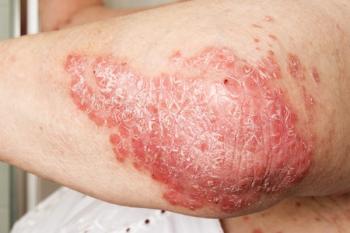
Current joint psoriasis guidelines from the American Academy of Dermatology and the National Psoriasis Foundation emphasize the need to help patients understand the relationship between psoriasis and comorbid conditions and the importance of seeking appropriate interventions as needed.

Eli Lilly announces the U.S. Food and Drug Administration approval of ixekizumab (Taltz) for treatment of moderate-to-severe plaque psoriasis in pediatric patients 6 and under 18 who are also eligible for phototherapy and systemic therapy.
Stem cell factor, a growth factor critical for melanocyte survival, may play an important role in the development of benign and malignant skin hyperpigmentation disorders.

Chemical peels offer important advantages for treating melasma. Researchers offer insights from a review of recent advances in chemical peel treatments in melasma and acne.

An expert speaks with Dermatology Times about the nuances of diagnosing, treating and managing eczema in black patients.