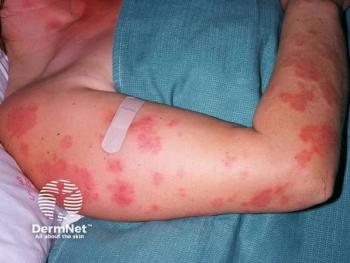
Tapinarof Cream 1% is FDA Approved
Dermavant’s new topical treatment for plaque psoriasis in adults was approved by the FDA this morning.
Tapinarof cream 1%, (Vtama; Dermavant Sciences) was approved by the FDA for topical treatment of plaque psoriasis in adults. This aryl hydrocarbon receptor agonist is the first steroid-free topical that is FDA approved in this class of treatments.
“Following 20-plus years of minimal innovation in the topical psoriasis treatment space, I believe the approval of Vtama cream is an important step in establishing a new treatment option for adults with mild, moderate and severe plaque psoriasis,” said Mark Lebwohl, MD, FAAD, dean for clinical therapeutics and Waldman professor and chairman emeritus of the Kimberly and Eric J. Waldman Department of Dermatology, Icahn School of Medicine at Mount Sinai in New York, New York, and lead author of the Phase 3 studies of tapinarof cream, and a part of the Dermatology Times® editorial advisory board, in the press release. “As a clinician, I’m excited to finally have a versatile, once-daily, steroid-free topical treatment that is backed by extensive clinical trial data supporting its favorable safety and efficacy profile and a demonstrated remittive effect of approximately four months in patients off therapy.”
The approval was based on phase 3 data from PSOARING 1 (NCT03956355) and PSOARING 2 (NCT03983980) clinical trials, both published in the New England Journal of Medicine.
According to the press release, more than 40% of phase 3 study patients (n=312/763) achieved complete disease clearance or physician global assessment (PGA=0) at least once during the study period. “For patients randomized to tapinarof cream in PSOARING 1 and PSOARING 2 who achieved a PGA of 0 during the 12-week study and subsequently enrolled in the Phase 3 LTE study (n=73), tapinarof cream demonstrated a remittive effect (maintenance of PGA of 0 or 1 while off therapy) with a median duration to first worsening of approximately four months,” the release continued. For the patients who entered the study with a PGA of 0, the mean remittive effect of off-therapy was 130 days. The safety profile was consistent through all of the trials.
There were responses taken after the PSOARING 3 trial, which found:
- Patients preferred tapinarof to prior topical treatments with 81.7% considering it more effective.
- 85.8% either strongly agreed or agreed they could easily manage their psoriasis with tapinarof.
- 82.5% expressed that they would use tapinarof again or continue using it if available.
The following pictures were supplied by Dermavant Sciences:
Reference:
FDA approves Dermavant’s Vtama (Tapinarof) cream, 1% for the treatment of plaque psoriasis in adults: first topical novel chemical entity launched for psoriasis in the US In 25 years. GlobeNewswire News Room. Published May 24, 2022. Accessed May 24, 2022. https://www.globenewswire.com/news-release/2022/05/24/2449068/34323/en/FDA-Approves-Dermavant-s-VTAMA-tapinarof-cream-1-for-the-Treatment-of-Plaque-Psoriasis-in-Adults-First-Topical-Novel-Chemical-Entity-Launched-for-Psoriasis-in-the-U-S-in-25-Years.html
Newsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.