Skin metagenomics reveals acne insights
A better understanding of the delicate balance the bacterial microbiome plays in disease may lead to new therapeutic options.
There has been a revolution in the last decade in the understanding of the skin’s microbiome and how organisms previously believed to be commensal actually play roles in skin diseases, such as
READ:
“In the old days, we used to culture skin to see what was growing…,” said Sheila Fallon Friedlander, M.D., professor of dermatology and pediatrics, University of California San Diego and Rady Children’s Hospital, and chair of the session Bacterial Infections and Skin-Staph, Strep and More. She spoke at the World Congress, in Vancouver, Canada (June 2015).
But that would only tell part of the story because certain bacteria and fungi are more likely to grow and reveal themselves in a culture. Others, such as Pityrosporum, will not grow on standard fungal culture medium, so if the dermatologist doesn’t ask the lab to apply olive oil or special nutrients, he or she wouldn’t know if it were a major inhabitant of a tested area. In addition, routine bacterial and fungal cultures may indicate that certain classes of organisms are present, such as Staph, but don’t reveal particular strains. It appears that certain strains can cause more pathology, Dr. Friedlander says.
Fortunately there is now a tool available to help dermatologists and others better identify particular strains of bacteria and fungi, according to Dr. Friedlander.
READ:
“Now, we have the ability to do metagenomics,” she says. “Metagenomics is a culture-free sequence-based method we can utilize to collect, analyze and understand the skin microbiome.”
Adult, neonatal acne focuses
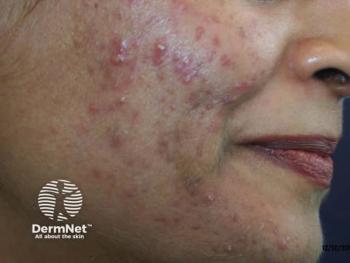
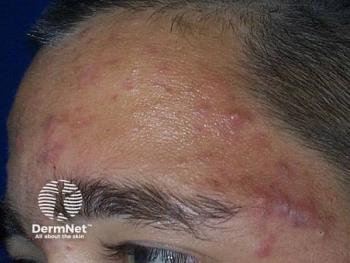
Taking a closer look at the skin reveals important information about bacteria, including strain types. And, when it comes to acne, those different strains can determine whether the presence of Propionibacterium acnes (P. acnes) results in acne or healthy skin.
READ:
“We used to say, acne in adults is caused by overgrowth of Propionibacterium acnes. Now we know that’s not the whole story. We think disease relates to a combination of the way the body’s immune system responds to the bacteria present in the pilosebaceous unit, as well as the hormones in your system and the responsiveness of the pilosebaceous unit to these hormones-particularly, androgen and its biological relatives,” Dr. Friedlander says. “They’re all playing a role.”
Researchers have found that P. acnes comprises about 87 percent of the bacteria living in the pilosebaceous unit. But its presence there doesn’t necessarily mean that a patient is going to have bad acne.
“What metagenomics has allowed us to do is rather than just culturing to see if there’s P. acnes, we’re now able to look at the bacteria genetically. We’re able to take samples from skin and then by doing … DNA amplification and sequencing, we can figure out what bugs are there and the strains of those bugs,” she says.
Specifically, researchers have identified P. acnes’ strains RT4 and RT5 as associated with acne. Whereas, if metagenomics finds more of the RT6 strain, that’s an indication that the skin is healthy, according to Dr. Friedlander.
READ:
“This shows the power of using these new tools-using DNA amplification and sequencing-to perform metagenomics,” Dr. Friedlander says. “We think these different strains may have relatively different invasiveness and relatively different effects on the immune response of the body. It may lead us to a whole different understanding of acne. Some strains may be good to have around, while others will lead to disease.”
The thinking also applies to neonatal acne, according to Dr. Friedlander.
“We think that neonatal acne probably bears a relationship to Malassezia furfur, which is a yeast often referred to as Pityrosporum that lives as a commensal on normal skin. But, in some cases, it becomes pathogenic or invasive,” she says. “There is an association in neonatal acne that has been shown in many studies [in] the presence of Malassezia. However, not all studies are clear-cut. We hypothesize that it may be particular strains that cause disease, or perhaps the infant’s innate immune response that may be differentially up-regulated in some children and not others.”
Metagenomics beyond acne
Metagenomics is changing the way researchers think about Staph epidermidis.
In the past, dermatologists might culture a child’s skin and be concerned about finding Staph aureus, but be reassured if Staph epidermidis grows, because that organism has always been thought to be a “good skin neighbor or resident,” according to the dermatologist.
READ:
“We’d say it doesn’t really matter that it’s there. It’s just sitting around,” Dr. Friedlander says. “Well, Rich Gallo and others have published studies which demonstrate that Staph epidermidis could play a very important role in the natural health of the skin. These organisms possess and/or elaborate substances which stimulate cellular activity, including skin-related immune response, so the skin is better able to fight off true pathogens.”
In addition, there are two skin disorders that have been associated with yeast infections: seborrheic dermatitis and tinea versicolor. Both are thought to be associated with Pityrosporum or Malassezia-yeast everyone has on their skin.
“In some people, the Pityrosporum becomes more pathogenic, and there are some circumstances that make people more high risk, including if they’re immunosuppressed, if they’re on long-term antibiotics or if they’re in a very humid environment,” Dr. Friedlander says.
This offers yet another example of the microbiome-organisms that are living on the skin that usually are not of consequence. These organisms may be doing something good for the body’s system but can turn against it.
READ:
“Perhaps, it also relates to the strain of Pityrosporum that predominates, and perhaps humidity and immunosuppression favor overgrowth of the deleterious strains. We don’t know yet, but metagenomics may help lead us to the answer,” Dr. Friedlander says.
How might this all play out?
Metagenomics could lead to a better understanding of which therapeutic options are most important.
In essence, dermatologists treating acne patients in the future might encourage growth of good P. acnes or other helpful bacteria, rather than concentrating on eradicating all P. acnes.
READ:
“I can see a future where it may be that we will identify ‘helpful’ bacterial and yeast strains. And when we’re treating disease, we might transplant beneficial bacteria from one part of the body to another, or more practically, apply a cream laden with ‘good bugs’ to the acne-prone areas,” according to Dr. Friedlander.
Pseudomembranous colitis patients are already receiving this type of treatment, with fecal transplants. Pseudomembranous colitis is the result of overgrowth of clostridium difficile in the gut, which induces life-threatening diarrhea. Health care providers can now transplant feces from healthy patients into those suffering from disease.
“It’s a situation where many of these organisms that we thought in the old days were just ‘hangers-on,’ may play a very important role in the proper stimulation of our immune system and the well-being of our skin,” she says.
To learn more on this topic:
Fitz-Gibbon S, Tomida S, Chiu B-H, et al.
Cogen AL, Yamasaki K, Sanchez KM, Dorschner RA, Lai Y, MacLeod DT, Torpey JW, Otto M, Nizet V, Kim JE, Gallo RL. Selective antimicrobial action is provided by phenol-soluble modulins derived from Staphylococcus epidermidis, a normal resident of the skin. J Invest Dermatol. 2010 Jan;130(1):192-200. http://www.ncbi.nlm.nih.gov/pubmed/19710683