Novel acne gel offers more tolerable treatment option
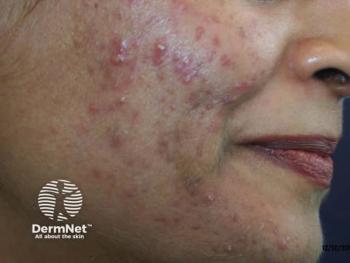
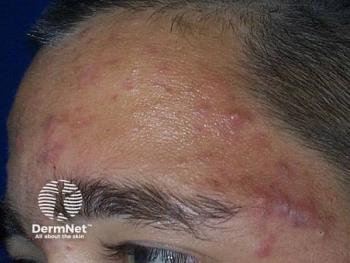
A novel gel formulation combining tretinoin 0.04% and clindamycin 1% using a controlled release microsphere delivery system effectively treats acne vulgaris without the skin irritation associated with topical tretinoin alone, according to recent data.
A novel gel formulation combining tretinoin 0.04% and clindamycin 1% using a controlled release microsphere delivery system effectively treats acne vulgaris without the skin irritation associated with topical tretinoin alone, according to results from a phase 3 trial published February 5 in the Journal of Dermatological Treatment.
Topical tretinoin is a proven acne treatment, but many patients have difficulty tolerating treatment-related skin irritation and sun sensitivity. Researchers studied use of a topical formulation with tretinoin microsphere (0.04%) and clindamycin (1%), comparing it to one of two monotherapies, tretinoin 0.025% or clindamycin 1%, in a study of 750 acne vulgaris patients.
“Tretinoin gel microsphere (0.04%) is a novel formulation developed to minimize tretinoin-associated cutaneous irritation while maintaining the efficacy. in this novel formulation, tretinoin is entrapped in porous polymeric microspheres that serve as a reservoir and deliver the drug in a controlled manner of a longer period, and thus, reducing irritation,” according to the authors.
The microsphere formulation also helps protect against tretinoin-related photo degradation, they write.
Researchers in India studied patients ages 12 years and older who had been diagnosed with facial acne. In the multicenter study, patients were randomized to receive one of the three therapies, applying it once daily for 12 weeks. All patients had an investigator’s Static Global Assessment (ISGA) score of 3 (moderate) or 4 (severe) at the study’s start.
They found that of the 300 patients who received the combination topical, the average inflammatory lesion count went from 29.42 at baseline to 6.88 at 12 weeks, compared to 29.93 average inflammatory lesion count at baseline to 8.87 at 12 weeks in the tretinoin group and 29.83 reduced to 10.28 in the clindamycin group. Noninflammatory lesion counts went from an average 38.28 to 11.62 at 12 weeks in the tretinoin and clindamycin treated patients, versus from 39.01 to 12.64 in the tretinoin group and 38.38 to 16.97 in the clindamycin group.
Among patients treated with the combination therapy, inflammatory lesions decreased 77%, noninflammatory 71% and total lesions 73% in 12 weeks of topical, once daily treatment. That was a significantly greater reduction than that of the 300 patients treated with tretinoin or 150 patients treated with clindamycin monotherapy.
Among other findings: At 12 weeks in the combination therapy group, five patients had a grade 4 ISGA score versus 38 at baseline. Forty-six percent of patients in the combination group achieved 2 or higher grade improvements.
While all three treatments were generally well tolerated, there were 20 adverse events reported. Six occurred in patients on the combination therapy, 11 on tretinoin monotherapy and 3 were in the clindamycin group. Nineteen of the 20 adverse events were mild-to-moderate and one, in the tretinoin group, was severe. None of the patients in the combination therapy group discontinued treatment because of adverse events during the 12-week study but two of the 300 patients in the tretinoin group discontinued treatment because of excessive irritation and increased acne lesions.
“The microsphere formulation facilitates controlled release of the drug; thereby, reducing the possibility of irritation and permitting the use of higher concentration of the drug for improved efficacy,” the authors write.
Disclosures:
Dr. Reddy’s Laboratories funded the study and company employees were among the authors.
References:
Dogra S, Sumathy TK, Nayak C, et al. Efficacy and safety comparison of combination of 0.04% tretinoin microspheres plus 1% clindamycin versus their monotherapy in patients with acne vulgaris: a phase 3, randomized, double-blind study. J Dermatolog Treat. 2020;:1-9.