FDA Grants Priority Review to Spesolimab
The FDA grants Priority Review for spesolimab (BI655130; Boehringer Ingelheim) as a treatment for generalized pustular psoriasis.
Boehringer Ingelheim today announced that the FDA has accepted a Biologics License Application (BLA) and granted Priority Review for spesolimab (BI655130; Boehringer Ingelheim), a humanized monoclonal antibody that targets interleukin (IL)-36, for the treatment of generalized pustular psoriasis (GPP) flares.1 This would be a first in class drug, which means that the medication uses a new and unique mechanism of action for treating a medical condition.
The FDA grants Priority Review to applications for medications that, if approved, would offer significant improvement over available options in the safety or effectiveness of the treatment, diagnosis, or prevention of serious conditions.
The FDA has granted spesolimab both an Orphan Drug Designation (ODD) for the treatment of GPP, and Breakthrough Therapy Designation (BTD) for spesolimab for the treatment of GPP flares in adults.
"The FDA acceptance of our filing for spesolimab is a critical step in our efforts to bring this first-in-class treatment to people living with GPP," said Matt Frankel, MD, vice president of clinical development and medical affairs, specialty care, with Boehringer Ingelheim in Ridgefield, Connecticut. “There is an urgent unmet need for an approved treatment option that can rapidly clear painful GPP flares."
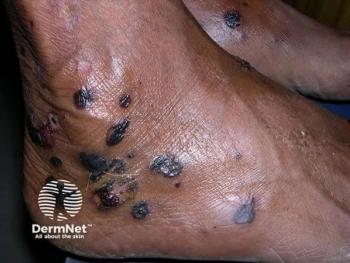
GPP is a rare, life-threatening neutrophilic skin disease, which is distinct from plaque psoriasis. It is characterized by episodes of widespread eruptions of painful, sterile pustules (blisters of noninfectious pus). Currently, there is an unmet need for treatments that can rapidly and completely resolve the signs and symptoms of GPP flares, which is what this treatment aims to do. Flares greatly affect a person’s quality of life and can lead to hospitalization with serious complications, including heart failure, renal failure, sepsis, and death,
Reference:
1. U.S. FDA grants Priority Review for spesolimab for the treatment of flares in patients with generalized pustular psoriasis (GPP), a rare, life-threatening skin disease. Accessed December 15, 2021. https://www.boehringer-ingelheim.us/press-release/us-fda-grants-priority-review-spesolimab-treatment-flares-patients-generalized