Skin Cancer
Latest News
Latest Videos

CME Content
More News

The FDA cited concerns about patient population heterogeneity and confirmatory trial design.

Nemvaleukin alfa produced a disease control rate of 50% in advanced melanoma and RCC.

The test now provides 3 validated insights: risk of metastasis, response to ART, and risk of recurrence.

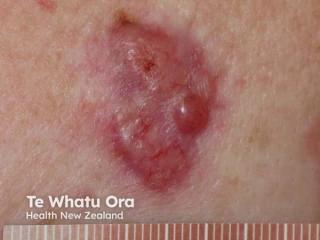
A case report presents a unique case of Bowen's disease affecting the lower extremities in a 75-year-old woman, which was initially misdiagnosed as a fungal infection.

At DERM 2025, David Cotter, MD, PhD, shared how GEP testing revolutionizes skin cancer management and enhances patient care at the forefront of dermatology innovation.

Irina Mazur spoke to Dermatology Times about the latest partnership, which aims to bridge the gap between clinical advice and patient behavior through personalized sun protection guidance.

Merck's pembrolizumab gains Health Canada approval for innovative perioperative treatment of advanced head and neck squamous cell carcinoma, enhancing patient outcomes.

Immutep advances eftilagimod alfa for head and neck squamous cell carcinoma, targeting patients with low PD-L1 expression and limited treatment options.
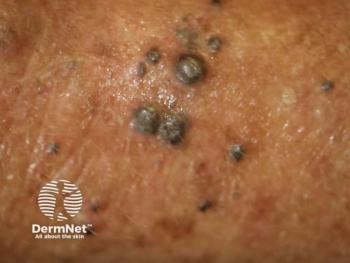
New clinical trial results reveal that intratumoral vidutolimod combined with pembrolizumab shows promise for treating advanced melanoma resistant to PD-1 therapy.

Kolansky identifies oxybenzone and fragrances as leading causes of sunscreen allergies.
Whitney Hovenic, MD's new SPOOGE sunscreen hopes to engage Gen Z in sun protection, merging skincare with fun and effective sun care strategies.

Discover how the Kimberly and Eric J. Waldman Melanoma and Skin Cancer Center at Mount Sinai leads in innovative skin cancer care and research, celebrating a decade of excellence.

Renata Block, DMSc, MMS, PA-C, interviews Adam Friedman, MD, FAAD, about his research into cannabidiol’s photoprotective potential, clinical trial design, and future directions.

Whitney Hovenic, MD, highlights alarming trends in Gen Z's sun protection habits and introduces SPOOGE, a new sunscreen brand promoting skin health.

At Elevate-Derm Summer, Buchi Neita, MCMSc, PA-C, discussed enhancing dermatologic care for patients with skin of color by recognizing characteristics in common conditions and offered strategies to build rapport with patients.

Discover how gene expression profiling transforms melanoma and SCC management, enhancing personalized treatment strategies and improving patient outcomes in skin cancer care.
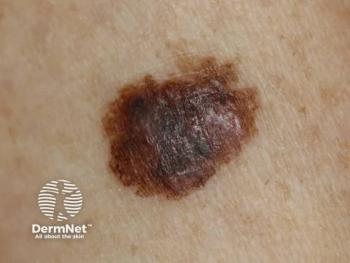
Castle Biosciences’ DecisionDx-Melanoma test plays a role in melanoma management with precise risk stratification, enhancing patient outcomes and reducing unnecessary surgeries.

Replimune faces FDA challenges for RP1's approval in advanced melanoma, highlighting complexities in cancer drug development and regulatory scrutiny.

This review of the latest dermatologic studies includes insights into ocular safety during laser procedures, radiation dermatitis prevention and management, and more.

At a recent Dermatology Times Case-Based Roundtable event, Gaurav Singh, MD, MPH, FAAD, FACMS, utilized 3 patient cases to explore the role of GEP testing in melanoma and SCC.

This review of the latest dermatologic studies includes insights into immunomodulatory effects of photodynamic therapy for skin cancer, in vivo and ex vivo sonographic evaluation of tumor margins, and more.

Artificial Intelligence (AI) enhances early detection of high-risk cutaneous squamous cell carcinoma (cSCC) by integrating advanced histopathological assessments for improved patient outcomes.

E-learning enhances skin cancer detection by training professionals in optical coherence tomography (OCT) for accurate differentiation between basal cell carcinoma (BCC) and non-BCC lesions.

At a recent Dermatology Times Case-Based Roundtable event, Andrew Baker, PA-C, MBA, explored the management of skin cancers with GEP testing through 4 complex cases.

Discover essential sun protection strategies from Dr. Susan Taylor, focusing on the impacts of UV, visible light, and personalized sunscreen choices.