Novel Glacial Rx Earns Third FDA Indication
R2 Technologies’ Novel Glacial Rx system has been cleared by the FDA for a new indication.
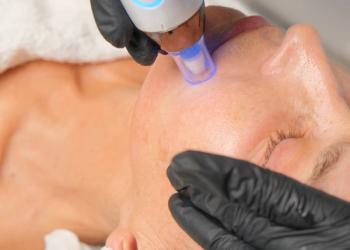
R2 Technologies Inc announced its Glacial Rx, which was FDA-cleared for general dermabrasion, scar and acne scar revision, and tattoo removal.1 It also now has an FDA product code. This indication is in addition to its already FDA cleared indications of removing benign lesions and reducing pain, swelling, and inflammation.
Tim Holt, the CEO of R2 Technologies, San Ramon, California, said that the FDA clearance was a “huge win” and that this is further proof that the product is so novel, that it warrants a new FDA category.
“R2 has been at the forefront of the med-esthetics space since inception with its unmatched technology and continues to lead advancements in the industry. We are excited to see the company break boundaries yet again with its latest indication,” said both David Present, MD, and Cherine Eldumiati Plumaker, R2 Technologies’ chairman and vice chairman respectively, San Ramon, California.
R2 Technologies has also established a new scientific advisory board that includes: Rox Anderson, MD, dermatologist from Boston, Massachusetts; Dieter Manstein, MD, PhD, dermatologist, from Boston Massachusetts; and Henry Chan, MD, PhD, dermatologist, and researcher, Boston, Massachusetts.
“With the appointment of our new Scientific Advisory Board, R2 is tapping into years of knowledge and expertise to launch new groundbreaking innovations, develop a superior product pipeline, and leverage technical insights to secure new clinical indications and expansion opportunities,” Holt said.
Reference:
1. R2 technologies’ Novel Glacial Rx system earns third FDA indication and unique product code. News Direct. Published September 28, 2021. Accessed September 28, 2021. https://newsdirect.com/news/r2-technologies-novel-glacial-rx-r-system-earns-third-fda-indication-and-unique-product-code-243799026
Newsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.