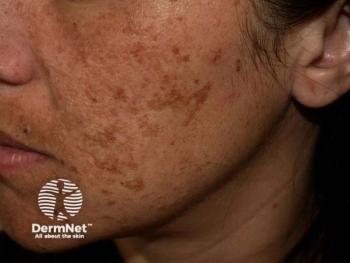
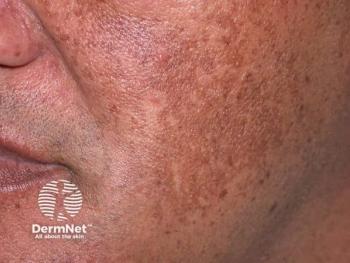
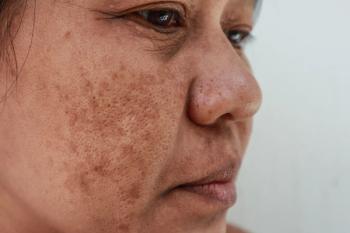
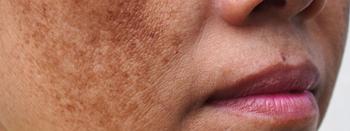
Investigators involved in a recent study1 found that a New Trio (NT) combination cream containing isobutylamido-thiazolyl-resorcinol, retinoic acid, and dexamethazone was efficacious in the treatment of melasma and may serve as an alternative to the melasma gold-standard treatment, known as Kligman’s Trio (KT).
The study, published in the Journal of the European Academy of Dermatology and Venereology, sought to compare the efficacy of the novel NT cream versus KT cream. According to study authors Bertold et al, KT’s efficacy in melasma has yet to be matched by alternative therapies.
The controlled, double-blinded, randomized, and prospective study was conducted from November 2021 to September 2022 in a single study center in France. Study participants (n=39) were adult patients who had received a melasma diagnosis via Wood’s light examination. Patients who were pregnant or breastfeeding or who had another facial pigmentary disorder were excluded from participation. Additionally, any patients who had a history of treatment with non-steroid anti-inflammatory therapeutics, depigmenting cosmetics, or corticosteroids (inhaled, systemic, or topical) on the face or eyes were also excluded.
Upon inclusion, patients were randomized to 2 treatment groups, one of which received NT cream while the other received KT cream. Both combination creams included 0.1% retinoic acid and 0.1% dexamethasone; however, NT cream contained 0.1% isobutylamido-thiazolyl-resorcinol, while KT cream contained 5% hydroquinone as its third ingredient.
Patients applied their assigned cream to their faces on a once-daily basis for a 12-week period. Identical topical sunscreens were distributed at baseline and applied in addition to the assigned combination cream, then with continued use through week 24 of the study. At inclusion, after the initial 12-week trial period, and again after the full 24 weeks of the study, a blinded dermatologist evaluated each participant in accordance with the Modified Melasma Area and Severity Index (mMASI), MelasQoL, Common Terminology Criteria for Adverse Events (CTCAE), and Visual Analog Scale (VAS).
Key Takeaways
- A prospective study compared the efficacy of New Trio (NT) combination cream and Kligman's Trio (KT) combination cream for melasma treatment, with NT demonstrating substantial improvement in Modified Melasma Area and Severity Index (mMASI) scores and quality of life (MelasQoL) scores.
- Patients in the NT treatment group experienced a 63% improvement in mMASI score between baseline and week 12, and an average MelasQoL score improvement of -12.57 at week 12, making NT a promising option for melasma treatment.
- The New Trio (NT) combination cream was found effective in treating melasma and as a potential alternative to Kligman's Trio (KT).
At baseline, week 12, and week 24, respectively, members of the NT treatment group had an average mMASI of 7.54 ±4.24, 2.93 ±4.09, and 4.40 ±4.35. At baseline, week 12, and week 24, members of the KT treatment group had an average mMASI of 6.08 ±3.31, 3.51 ±3.57, and 3.64 ±2.76. Between the assessments at baseline and week 12, patients in the NT group had an average 63% improvement in mMASI score, while patients in the KT group had an average mMASI score improvement of 39%.
At week 24, mMASI scores from both treatment arms had increased from week 12, which investigators attributed to relapse upon discontinuation of treatment.
Regarding quality of life, patients in the NT treatment group had a –12.57 average improvement in MelasQoL score at week 12 and an average improvement of –6.95 at week 24. Patients in the KT treatment group had a –6.66 average MelasQoL score improvement at week 12 and a –3.67 average improvement at week 24.
Of 97 reported adverse events, 3 were considered medically significant. However, these were deemed unrelated to treatment. Treatment-related adverse events included acne, desquamation, erythema, hypertrichosis, and irritation, with nearly equal incidence in both the NT group (n=36) and the KT group (n=38).
“Together these data attest that the New Trio combination demonstrated efficacy and significant improvement in the quality of life in treating melasma patients,” study authors wrote. “This new approach could be considered as an alternative to the Kligman’s Trio.”
Reference
- Bertold C, Fontas E, Singh T, et al. Efficacy and safety of a novel triple combination cream compared to Kligman’s trio for melasma: A 24‐week double‐blind prospective randomized controlled trial. J EurAcad Dermatol Venereol. Published online August 24, 2023. doi:10.1111/jdv.19455