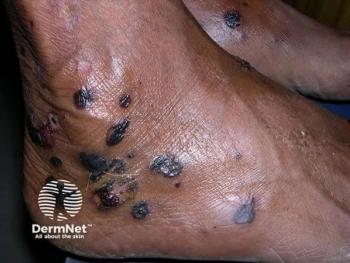
FDA Grants Priority Review for Dupilumab sBLA for Adults with Prurigo Nodularis
Dupilumab (Dupixent; Sanofi and Regeneron Pharmaceuticals, Inc) has received priority review for its supplemental Biologics License Application by the FDA to treat adult patients with prurigo nodularis.
Today it was announced that dupilumab (Dupixent; Sanofi and Regeneron Pharmaceuticals, Inc), an IL-4 and IL-13 inhibitor, has received the FDA’s approval of getting priority review for its supplemental Biologics License Application (sBLA) for the treatment of adult patients who have prurigo nodularis (PN). It will be the first and only medicine specifically indicated to treat PN, according to the release.
The data supporting the sBLA is from 2 phase 3 trials, LIBERTY-PN PRIME (NCT04183335) and PRIME2 (NCT04202679), which both investigating the safety and efficacy of dupilumab as a treatment in patients 18 years and older with uncontrolled PN.
In both trials, patients achieved the primary and key secondary endpoints which demonstrated the dupilumab improved disease symptoms, such as a reduction in itch and skin lesions, significantly compared to placebo. The press release stated that the safety profile of dupilumab in these trials and patient population were like those observed in the atopic dermatitis studies, with the most common adverse event being conjunctivitis.
The target action date for the FDA decision is September 30, 2022.
Reference:
Sanofi: Press Releases, Tuesday, May 31, 2022.