Amyrt Receives FDA Complete Response Letter for Oleogel-S10 to Treat Epidermolysis Bullosa
The company was asked by the FDA to submit additional confirmatory evidence of effectiveness in the treatment of epidermolysis bullosa.
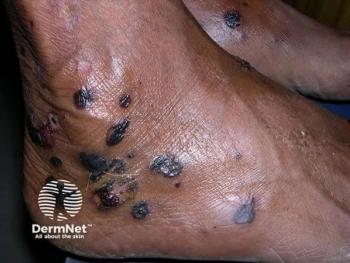
Amryt, a global, commercial-stage biopharmaceutical company dedicated to acquiring, developing, and commercializing novel treatments for rare diseases, announced it has received a complete response letter (CRL) from the FDA regarding its new drug application (NDA) for Oleogel-S10, for the treatment of the cutaneous manifestations of dystrophic and junctional epidermolysis bullosa (EB). EB is a rare, genetic skin disease characterized by extremely fragile skin that blisters and tears from minor friction or trauma for which there are no approved treatment options, according to the press released.
The FDA stated its review of the application was complete and determined the application cannot be approved in its present form. Amryt has been asked to submit additional confirmatory evidence of effectiveness for Oleogel-S10 in EB. According to the release, Amryt intends to discuss with the FDA the nature of the data required to address the agency’s concerns.
“We are extremely disappointed by this decision and we remain committed to our goal of bringing Oleogel-S10 to patients. EB is a devastating disease with no approved treatment options and the EB community is desperate for a therapy,” said Joe Wiley, CEO of Amryt Pharma. “The EASE study [NCT03068780]was the largest randomized clinical trial ever conducted in this disease and the first phase 3 study to meet its primary endpoint. We are committed to working collaboratively with the FDA to identify the most expeditious pathway towards a potential approval for this important therapy.”
Reference:
Amryt receives complete response letter from the FDA for Oleogel-s10 NDA. GlobeNewswire News Room. Press release. Published February 28, 2022. Accessed March 8, 2022. https://www.globenewswire.com/news-release/2022/02/28/2393303/0/en/Amryt-Receives-Complete-Response-Letter-from-the-FDA-for-Oleogel-S10-NDA.html