- Dermatology Times, April 2021 (Vol. 42, No. 4)
- Volume 42
- Issue 4
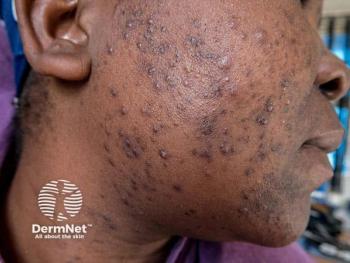
Tazarotene Excels Across BMI Categories
Data from trials show tazarotene 0.045% lotion is an effective treatment for moderate to severe facial acne and improves acne-related QOL in patients across BMI categories.
A post hoc analysis of data from the pivotal trials investigating tazarotene 0.045% lotion (Arazlo; Ortho Dermatologics) for treating moderate to severe facial acne show the efficacy, safety, and tolerability of the novel topical retinoid formulation are robust irrespective of patient body mass index (BMI). In addition, patients in all BMI categories and particularly those who are overweight or obese benefit with improvements in acne-related quality-of-life measures.
With research presented as a poster at Maui Derm 2021,1 Fran E. Cook- Bolden, MD, says the findings are important considering the growing population of overweight and obese individuals in the United States and the negative impact of both high BMI and acne on health-related quality of life.
“Results of a recent systematic review found that the prevalence of acne is higher in overweight and obese individuals compared with those in the underweight/normal weight BMI category,” according to Cook-Bolden, who is director of the Skin Specialty Dermatology and the Ethnic Skin Specialty Group at Park South Medical in the Bronx, New York, and assistant clinical professor of dermatology at Columbia University in New York, New York. “However, studies examining the potential for BMI-related differences in quality of life among patients with acne are lacking. The effect of BMI on the efficacy and safety of topical acne treatments is largely unknown. “Patients in the tazarotene pivotal trials represented a diverse population with respect to BMI and other demographic characteristics. Based on the results of this post hoc analysis, physicians can feel confident prescribing tazarotene 0.045% lotion for their acne patients, no matter their BMI,” says Cook-Bolden.
Cook-Bolden was an investigator in the tazarotene 0.045% lotion clinical trials that enrolled patients 9 years and older with moderate-to-severe acne (Evaluator’s Global Severity Score [EGSS] of 3-4) and randomized them 1:1 to treatment with either topical medication or vehicle once daily for 12 weeks. A total of 1612 patients were included in this post hoc analysis, of whom 903 were categorized as underweight/normal weight (BMI < 25 kg/m2), 377 as overweight (BMI 25 kg/m2 to < 30 kg/m2) and 332 as obese (BMI ≥ 30 kg/m2). Compared with the lowest BMI category, the overweight and obese cohorts included higher proportions of female, Black, and Hispanic patients.
The potential for BMI-related effects on treatment response was investigated using a variety of efficacy end points, including percentage reduction in inflammatory lesions, percentage reduction in noninflammatory lesions, percentage of participants achieving treatment success (≥ 2-grade improvement from baseline in EGSS and a rating of “clear” or “almost clear”), and improvements in the Acne-Specific Quality of Life questionnaire.
In all BMI groups, patients treated with tazarotene achieved approximately a 55% to 60% reduction from baseline to week 12 in inflammatory and noninflammatory lesion counts. The difference between the tazarotene- and vehicle-treated groups was statistically significant for both end points in all BMI subgroups—except that the magnitude of reduction in inflammatory lesions was similar in the vehicle and tazarotene groups among the obese patients.
Across the 3 BMI subgroups, statistically significant differences favoring tazarotene over vehicle were observed in comparisons of treatment success rates. Treatment success rates for tazarotene-treated patients ranged from approximately 29% to 34% across the 3 BMI subgroups compared with approximately 16% to 22% for vehicle-treated patients.
Data from the quality-of-life questionnaire showed improvements in scores for all 4 domains (self-perception, role-emotional, role-social, and acne symptoms) with both vehicle and tazarotene in all BMI subgroups. In most comparisons, however, the change from baseline was greater in the tazarotene-treated patients compared with the vehicle group. The improvements in quality of life also tended to be higher in the overweight and obese patients than in those with a BMI less than 25 kg/m2.
“In the pivotal trials investigating tazarotene 0.045% lotion, patients in the overweight and obese BMI subgroups had numerically worse quality of life at baseline than patients in the lowest BMI group,” Cook-Bolden notes. “Findings of a previous analysis of the pooled pivotal trial data showed that participants with worse quality of life at baseline had greater improvements in quality of life at the end of the study.3
“A previous analysis also found that quality-of-life improvements were greater among female versus male and Black versus White patients.3 It’s interesting that evidence exists showing that [female patients] with acne are more likely than [male patients] to develop anxiety/depression because of their condition and that acne improvement positively affects quality of life for [female patients].”4
Adverse event and cutaneous safety and tolerability data were also analyzed with patients stratified by BMI; the results showed no evidence that the safety and tolerability profile of tazarotene differed among the BMI subgroups.
Disclosures:
Cook-Bolden has served as consultant, speaker, investigator for Galderma, LEO Pharma, Almirall SA, Cassiopea SpA, and Ortho Dermatologics, and as an investigator for Encore, Foamix, Hovione, Aclaris Therapeutics Inc, and Cutanea.
References:
- Cook-Bolden FE, Green L, Keri J, et al. Impact of body mass index on efficacy, safety, and tolerability of once-daily tazarotene 0.045% lotion in patients with moderate-to-severe acne. Poster presented at: Maui Derm Live In-Person Dermatology CME Conference and Maui Derm Connect Virtual Dermatology CME Conference; January 25-29, 2021; Maui, Hawaii; virtual.
- Heng AHS, Chew FT. Systematic review of the epidemiology of acne vulgaris. Sci Rep. 2020;10(1):5754. doi:10.1038/ s41598-020-62715-3
- Kircik LH, Lain E, Gold M, et al. Effects of tazarotene 0.045% lotion on quality of life in patients with moderate-to-severe acne. J Drugs Dermatol. 2020;19(11):1086-1092. doi:10.36849/JDD.2020.5457
- Skroza N, Tolino E, Proietti I, et al. Women and acne: any difference from males? a review of the literature. G Ital Dermatol Vene- reol. 2016;151(1):87-92.
Articles in this issue
over 4 years ago
Regenerative Medicine Gaining Popularityover 4 years ago
Combination Foam May Benefit Patients of Coloralmost 5 years ago
AI Diagnostics Fall Short in Skin of Coloralmost 5 years ago
Microneedling Strategiesalmost 5 years ago
Neuromodulators Combat Key Rosacea Challengesalmost 5 years ago
Baricitinib Demonstrates Positive Outcomes for Alopecia Areataalmost 5 years ago
COVID-19 Vaccine Refusal: Grounds for Termination?almost 5 years ago
Solving Pierced Earring Challengesalmost 5 years ago
Emerging Agents Augment Melasma Modalitiesalmost 5 years ago
Laser Device Purchases Are Guided by Multiple FactorsNewsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.