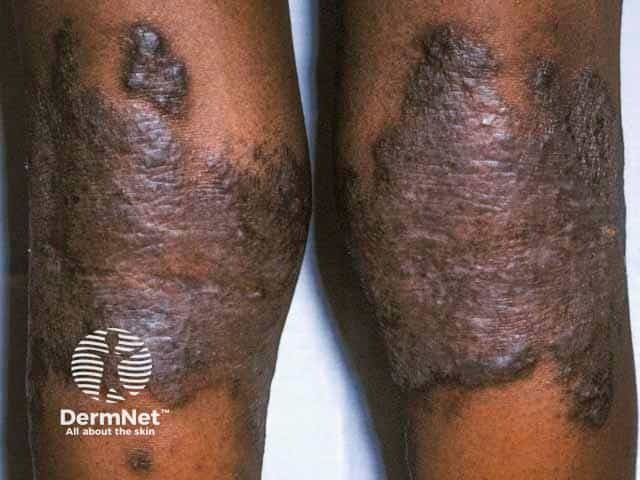
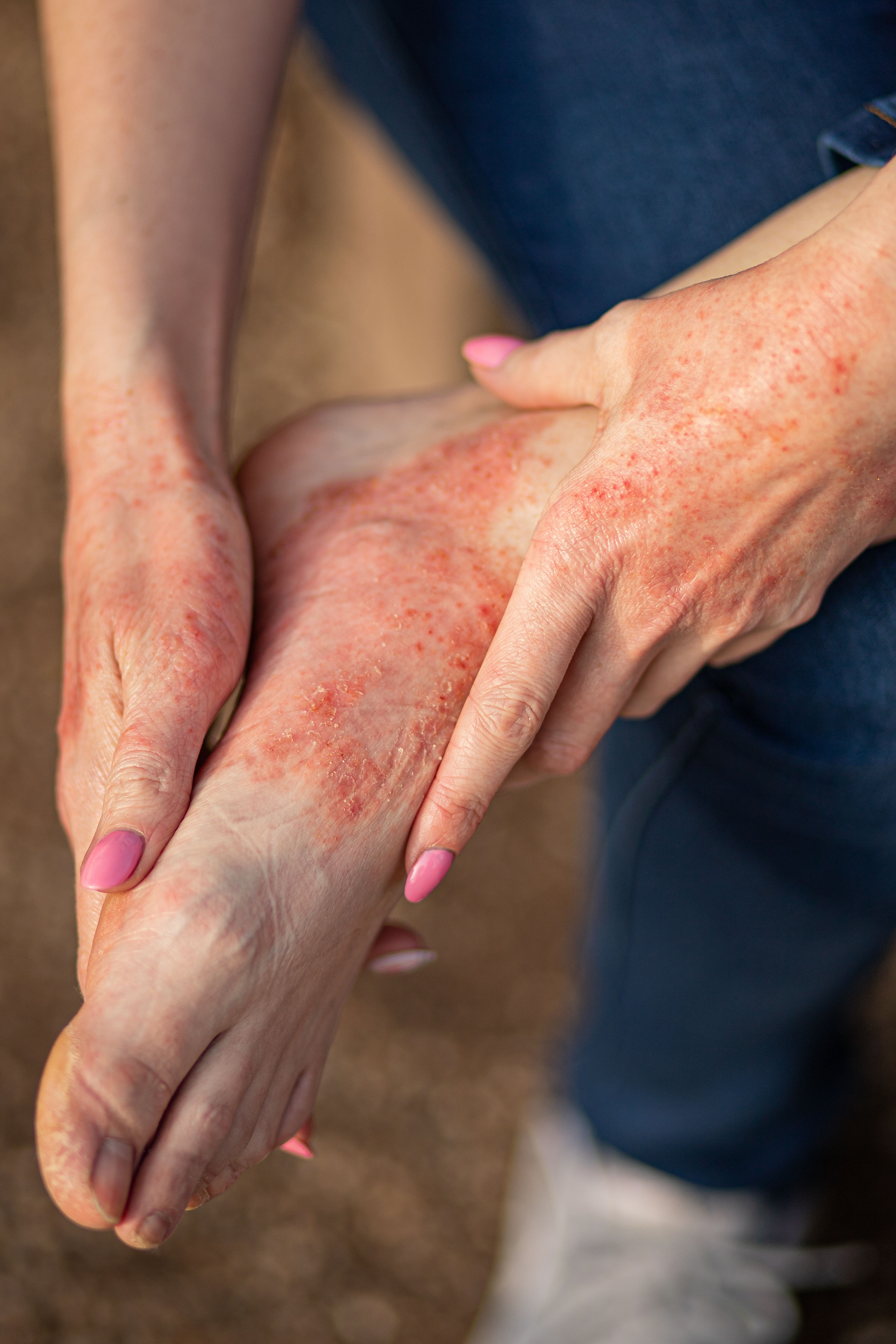
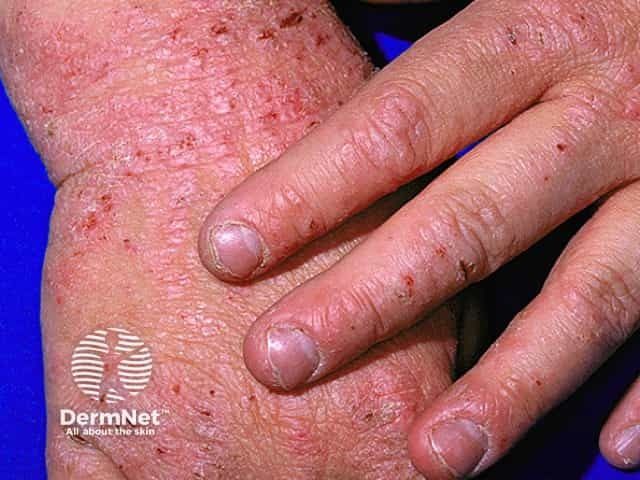
Atopic Dermatitis
Latest News
Latest Videos

Shorts




CME Content
More News
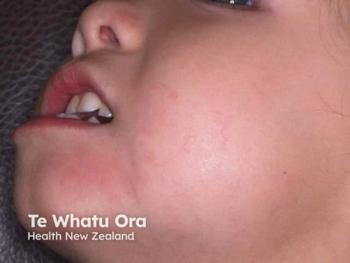
With 80% of trial participants being children, this analysis provides some of the most robust early-response data available for a non-steroidal topical in pediatric AD.
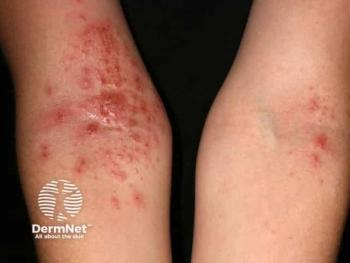
Targeted therapies are increasingly used earlier in treatment rather than being reserved for severe or refractory cases.
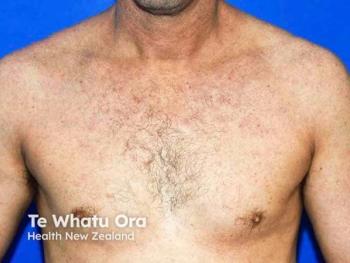
Phase 2 APEX part A data showed zumilokibart maintained EASI-75 responses in 75–85% of week 16 responders at 1 year across quarterly and biannual dosing regimens.
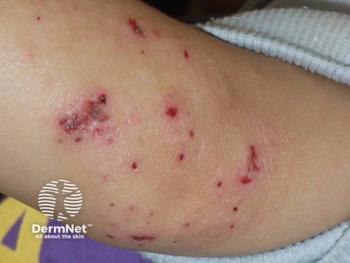
New data show that nearly 40% of eczema patients face insurance-related barriers, delaying or preventing access to prescribed therapies.

Meta-analysis finds sexual dysfunction common in asthma and AD, with rates hitting 54% in asthma, prompting better screening.
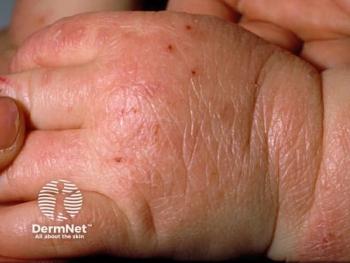
Positive phase 3 findings from ADorable-1 suggest lebrikizumab could expand biologic treatment options for younger patients with atopic dermatitis (AD).

Douglas DiRuggiero, DMSc, MHS, PA-C, reviewed clinical cases highlighting the distinct roles of topical ruxolitinib, tapinarof, and roflumilast in managing itch, inflammation, and long-term disease control in atopic dermatitis.

Omar Noor, MD, shares pearls on type 2 inflammation, including dupilumab insights for atopic dermatitis, prurigo nodularis, and CSU at Horizons in Advanced Practice.

Rocatinlimab had met primary end points in large phase 3 trials and was previously expected to move toward regulatory submission in 2026.
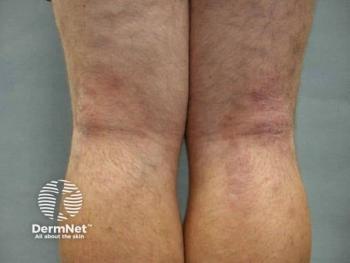
The double-blind, randomized, placebo-controlled ARQ-234-131 study will assess safety, tolerability, pharmacokinetics, and early signs of clinical activity.

At Winter Clinical Miami 2026, April Armstrong, MD, MPH, spotlights new pediatric topicals and JAK safety insights, plus targeted options for head-and-neck atopic dermatitis.

More than 57% of patients with ACD reported moderate to severe quality-of-life impairment, with symptoms and embarrassment among the most affected areas.

Castle Biosciences’ AdvanceAD-Tx test stratifies patients by molecular profile, identifying patients more likely to achieve faster and deeper responses with JAKs compared with Th2-targeted treatments.

A post hoc analysis found that abrocitinib provided faster itch relief than dupilumab in adults with itch-dominant atopic dermatitis.
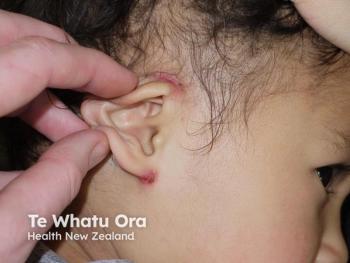
In pivotal studies, significantly more patients achieved clear or almost clear skin after 4 weeks of twice-daily difamilast compared with vehicle.

Targeting IL-4 and IL-13 signaling with dupilumab has demonstrated significant reductions in total and food-specific IgE in pediatric AD populations.

Investigators identified overlap between the blood immune profiles of patients with depression and inflammatory skin diseases, highlighting the Th2 axis as a potential treatment target.

The consensus summarizes contemporary real-world data showing that nearly 1 in 5 adolescents and adults with atopic dermatitis receive systemic corticosteroids.
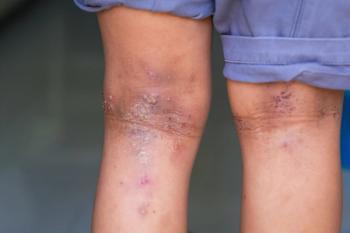
REZOLVE-AD shows rezpegaldesleukin delivers durable atopic dermatitis control with monthly or quarterly dosing, supporting Nektar’s phase 3 plans.

A new molecular test offers clinicians a way to personalize treatment for moderate to severe atopic dermatitis and reduce therapeutic cycling, helping to achieve faster control and a better experience for patients.

At MOPD 2026, Nanette Silverberg, MD, and Lisa Swanson, MD, unpack teen AD, fragrance allergy, and social media skin care hype harming barriers.

Arcutis reveals promising phase 2 results for roflumilast cream in infants with AD, highlighting safety and potential efficacy.

Kyowa Kirin takes full control of rocatinlimab, aiming to address unmet needs in atopic dermatitis while ensuring continuity in clinical trials.

A phase 2 trial reveals soficitinib's potential to significantly improve symptoms and quality of life in patients with moderate to severe AD.

Amlitelimab shows promise as a long-term therapy for atopic dermatitis (AD), offering effective results and favorable safety in recent clinical trials.