- Dermatology Times, March 2021 (Vol. 42, No. 3)
- Volume 42
- Issue 3
New E-BPO Improves Rosacea
This formulation could be first FDA-approved single-agent E-BPO prescription drug to treat rosacea.
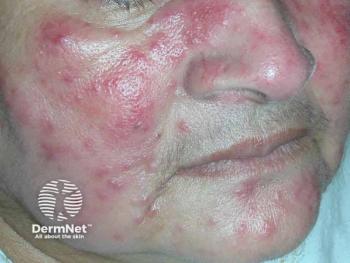
In a recent extension study of 2 phase 3 clinical trials, a novel 5% encapsulated benzoyl peroxide (E-BPO) cream (Epsolay, Sol-Gel Technologies Ltd) demonstrated significant superiority over vehicle in achieving clear or almost clear skin and reducing the number of lesions in patients with papulopustular rosacea. Data also showed improvement in facial erythema and telangiectasia.1
According to study authors, the primary objective of the extension study was safety evaluation. Findings showed the cream was well tolerated with adverse events (AEs) and cutaneous safety and tolerability comparable to the vehicle group.1 Results supported other research that found microencapsulation not only extends drug delivery time but improves efficacy and potently reduces the potential for skin irritation.2
“Epsolay uses a patented microencapsulation technology that effectively reduces the risk of skin irritation by encapsulating the BPO inside porous silica microcapsules,” said Neal Bhatia, MD, director of clinical dermatology, Therapeutics Clinical Research, San Diego, California, and lead author of the study. “The capsules form a barrier between the skin and the BPO crystals or other ingredients, allowing for the active drug to be released in a timely fashion, resulting in less skin irritation and a much higher tolerability and amenability of the medication in patients.”
The study results addressed a key challenge; the idea that BPO causes more skin issues. “The phase 3 trials were actually surprising in their outcome because the historical opinion was that BPO was too irritating for rosacea skin, which in the nonencapsulated form is true,” he said.
Philip Werschler, MD,FAAD, FAACS, assistant clinical professor of medicine/dermatology, University of Washington School of Medicine in Seattle, founding member of Spokane Dermatology Clinic, in Spokane, Washington, and coauthor of the study. “The Sol-Gel technology found in Epsolay was very well tolerated and very effective.”
Bhatia and Werschler presented a poster on the study findings at Maui Derm Live. The in-person dermatology continuing medical education (CME) conference in Hawaii was held concurrently with Maui Derm Connect, a virtual CME conference, from January 25 to January 29, 2021.1
DATA REVEAL FASTER OUTCOMES
Patients were followed for up to 40 additional weeks in the extension and for up to a total of 52 weeks, including the 12 weeks in the phase 3 trial.1
At each follow-up visit in the extension study occurring at baseline and at 4-week intervals out to week 40, patients were assessed using a 5-point IGA scale of rosacea severity and completed a Rosacea-specific Quality of Life index (RosaQoL) questionnaire. Outcome measures included an Investigator’s Global Assessment (IGA) status of “clear or almost clear”, number and timing of retreatments, and tolerability at 40 weeks. IGA success was defined as achievement of clear or almost clear at the 40-week follow-up visit.
These phase 3 trials (SGT 54-01 and SGT 54-02) evaluated the efficacy, safety, and tolerability of Epsolay in a total of 547 patients, 363 previously treated with Epsolay cream and 184 patients previously treated with vehicle. The study included male and female patients aged at least 18 years with moderate to severe rosacea with a baseline IGA score of 3 (moderate severity) or 4 (severe) on a severity scale of 0-4, between at least 15 and 70 total inflammatory lesions with 2 or fewer nodules present.1
Results showed a progressive improvement of rosacea symptoms over the total 52-week study period. At the 40-week follow-up, 67.2% of patients (66.5% in the vehicle group; 67% in the E-BPO group) achieved IGA success. In the extension study, there was an average of 1.4 retreatments with an average of 58 treatment-free days. The investigators also noted that facial erythema generally improved during the study. In addition, results from the RosaQoL questionnaire showed improvements from baseline to week 40 in the total score, symptom subscale score, functional subscale score, and emotional subscale score.1
For each of the cutaneous safety and tolerability parameters, data showed small increases in the percentages of patients with no or mild signs and symptoms over 52 weeks. At least 1 treatment-related AE was reported by 185 patients (34.6%), which were mild to moderate in severity and were not considered to be related to the study treatment. Ten patients experienced serious AEs, none of which were considered to be related to the study treatment.
“I think for those patients who have not had a good chance of recovery and good maintenance, Epsolay cream could do well for them because the activity of BPO in the study led to a quicker clearance in patients who applied the product,” Bhatia said.
THE FUTURE OF EPSOLAY
The FDA is currently evaluating Epsolay, but according to a press release, if approval is passed this spring, Sol-Gel Technologies Ltd projects that the novel formulation has the potential to be the first single-agent BPO prescription drug product.2
“This innovative formulation is coming out at a great time because many patients with rosacea have gotten frustrated with their existing therapies that often do not adequately address their symptoms,” Bhatia said. “I think Epsolay cream might be better for patients, particularly because it’s also cream based which might be more amenable to patients.”
Disclosures:
Bhatia and Werschler report no relevant or financial disclosures.
References:
1. Bhatia N, Werschler W, Baldwin H, et al. Long-term efficacy and safety of benzoyl peroxide cream, 5%, prepared with microencapsulation in papulopustular rosacea: results from an extension of two phase 3, vehicle-controlled trials. Poster presented at: Maui Derm Live In-Person Dermatology CME Conference and Maui Derm Connect Virtual Dermatology CME Conference; January 25-29, 2021; Maui, Hawaii; Virtual.
2. Erlich M, Arie T, Koifman N, Talmon Y. Structure elucidation of silica-based core-shell microencapsulated drugs for topical applications by cryogenic scanning electron microscopy. J Colloid Interface Sci. 2020;579:778-785.
Articles in this issue
about 5 years ago
2021’s Top Challenges Facing Physiciansabout 5 years ago
Optimize Risk, Management of Pigmented Lesions in Pediatric Patientsabout 5 years ago
Systemic Therapy Advances for ADabout 5 years ago
New Research Addresses Rosacea Challengesabout 5 years ago
Sebaceous Gland is Target for Acne Treatmentsabout 5 years ago
Quantifying is Key to Effective Hair Loss Treatmentabout 5 years ago
The Potential of Precision Medicineover 5 years ago
Novel Therapeutics Usher in a Year of Innovationover 5 years ago
Systemic Therapy Advances for Atopic Dermatitis