- Dermatology Times, May 2021 (Vol. 42, No. 5)
- Volume 42
- Issue 5
Medical, Surgical Tactics Help Manage HS
Thanks to new drug approvals, medical management is replacing surgery as the first-line treatment for inflammation caused by hidradenitis suppurativa. However, surgery is often necessary to relieve pain related to tunnels.
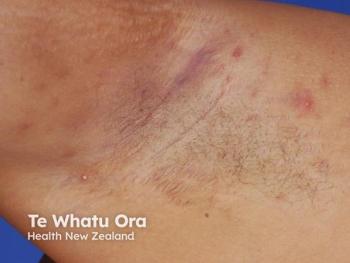
Over the past decade, the paradigm for treating hidradenitis suppurativa (HS) has shifted away from surgical management as the definitive treatment, to initially controlling the inflammatory component of this debilitating skin disease with medical therapies, such as broad-spectrum antibiotics and biologics, followed by surgery if needed.
“Once the inflammatory component of the disease is controlled, we are now advocating for limited surgery to manage and remove permanently disfigured or permanently scarred tissue that cannot be treated effectively with systemic medical therapies,” said Haley Naik, MD, an assistant professor of dermatology and director of the hidradenitis suppurativa program at the University of California, San Francisco.
Regardless, diagnostic delay is a huge challenge for the condition. “We medical professionals still have a lot of work to do to accurately diagnose HS patients early on in disease course when we potentially have the opportunity to alter disease trajectory,” said Naik, who led the symposium on medical and surgical management of HS at the American Academy of Dermatology (AAD) 2021 Virtual Meeting Experience (VMX) held April 23-25, 2021.1
The mainstay of current medical management for moderate to severe disease is tumor necrosis factor (TNF) inhibitors, such as adalimumab, which is currently the only FDA-approved treatment for HS.
Clinical trials (PIONEER I [
Other TNF inhibitors used to treat HS include infliximab (Avsola; Amgen) as well as hormonal treatments such as high estrogen-to-progesterone–ratio oral contraceptives and spironolactone.
In addition, systemic retinoids, particularly acitretin, can be used to treat patients with HS who have a prominent comedonal component to their disease.
“Despite a variety of available medical interventions, we are not yet at a stage with HS where there is a single silver-bullet treatment,” Naik told Dermatology Times®. “We are still layering therapies, so a patient may be on a biologic and an antibiotic, or on antibiotics and an agent to control hormonal fluctuations associated with their disease.”
For surgical management of HS, proper patient staging is crucial. “The most well recognized and easiest to use staging system is the Hurley clinical staging system, comprising 3 stages of disease,” Naik said.
Stage 1 disease consists of patients who develop intermittent nodules and abscesses without any associated scarring or tunnel formation. “These patients are treated almost exclusively with medical therapy: either systemic agents or, in some cases, topical agents,” Naik said.
Stage 2 disease is characterized by nodules and abscesses along with development of tunnels and scars. “Tunnels are the first sign of disease progression in HS,” Naik said. “Even a single tunnel is considered stage 2 disease.”
After optimizing inflammation control, these patients will usually require some type of localized surgery to remove tunnels, according to Naik, because tunnels cannot be treated with any available medical therapy. One highly effective localized surgical procedure that can be performed outpatient under local anesthesia is deroofing or unroofing, whereby the roofs of tunnels are removed and the base of the tunnels are cleaned and cauterized, she added.
“Patients are sent home with petrolatum dressing and a bandage and are asked to change their bandage once or twice a day,” Naik said. “The wounds heal by secondary intention. For limited lesions, complete healing typically occurs in 6 to 8 weeks. Patients are also still quite functional after the procedure despite having a surgical wound.”
These patients also report significant improvement in pain once the HS tunnels are removed. “Many patients report almost immediate pain relief after local anesthesia has worn off,” Naik said.
Hurley stage 3 disease is defined as nodules and abscesses with multiple interconnected tunnels and significant scarring, which often requires inpatient surgery under general anesthesia.
Wide, local excision is the preferred surgical treatment for these patients. “All of the damaged tissue in a given anatomical location is removed,” Naik said. “Surgeons will also often take an additional margin to ensure that all of the diseased tissue has been completely removed.”
A typically postoperative patient stay is 2 days for an uncomplicated case, with a recovery period for all patients lasting weeks to months.
“However, response rates are much more difficult to discern,” Naik said. “Response rates vary widely in part because there is no single uniform technique to perform these procedures. Fortunately, clarifying surgical definitions and procedures is an area of active investigation.”
Among the numerous promising targeted agents to manage HS are interleukin (IL)-17 and IL-23 inhibitors, as well as Janus kinase (JAK) inhibitors.
“These developments suggest that the future for HS medical management is quite bright,” Naik said.
Naik also noted the emphasis now on pain management for HS. “Pain is the most common symptom associated with the disease and the number 1 complaint of patients living with this condition,” Naik said. “We hypothesize that inflammatory and neuropathic mechanisms contribute to HS. The latter mechanism can be treated with gabapentin and doxepin.”
Disclosure:
Naik has received grant support from AbbVie; consulting fees from 23andMe, AbbVie, and DAVA Oncology; and advisory board fees from Boehringer Ingelheim. She is also an investigator for Pfizer, an associate editor for JAMA Dermatology, and an unpaid board member of the Hidradenitis Suppurativa Foundation.
References:
1. Naik H. Hidradenitis suppurativa/acne inversa (HS/AI): current medical and surgical management. Presented at: American Academy of Dermatology 2021 Virtual Meeting Experience (VMX) annual meeting; April 23-25, 2021.
2. Kimball AB, Okun MM, Williams DA, et al. Two phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375(5):422-434. doi:10.1056/NEJMoa1504370
Articles in this issue
about 5 years ago
Administrative Burden of Prescribing Biologicsabout 5 years ago
IL-36 Inhibitors on the Horizon for Generalized Pustular Psoriasisabout 5 years ago
Emerging Biologics Make Waves in ADabout 5 years ago
Master Techniques to Treat Eye Issuesabout 5 years ago
Current and Upcoming Topicals in the Pipeline for ADabout 5 years ago
I Used Botulinum Toxin Injections for Oily Skin—Now I Have Been Suedabout 5 years ago
Off-label Biologics Target Wide Range of Skin Diseasesabout 5 years ago
Debunking Sunscreen Controversiesabout 5 years ago
Off-label Sarecycline Combats Papulopustular Rosaceaabout 5 years ago
What’s Trending in Skin Care? May 2021