- Dermatology Times, October 2022 (Vol. 43. No. 10)
- Volume 43
- Issue 10
New Directions in the Treatment of Basal Cell Carcinoma
Two dermatologists discuss how immune checkpoint inhibitors will change the way dermatologists manage skin cancer.
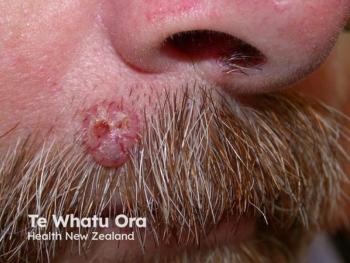
In the treatment of basal cell carcinoma (BCC), “The name of the game is identifying the lesions early on and then treating them accordingly, right? A lot of what I hear is, ‘Hey, doc, if it’s not going to kill me, it’s slow growing, why should I take care of it? Why should I bother doing anything?’ Then usually I show them one picture of what an advanced basal cell cancer can look like, and they’re ready to begin treatment,” said Sherrif F. Ibrahim, MD, PhD, an associate professor in the Department of Dermatology at the University of Rochester Medical Center in New York.
During a Dermatology Times® Recognize and Refer episode,1 Ibrahim and Vishal Patel, MD, FAAD, FACMS, BCC discussed a promising new therapy option: immune checkpoint inhibitors. Patel is an associate professor of dermatology at George Washington University School of Medicine and Health Sciences and director of the cutaneous oncology program at George Washington University Cancer Center in Washington, DC.
Ibrahim and Patel both noted that they’re seeing an increasing incidence of BCC, especially in younger patients. Patel explained that BCC is the most common type of skin cancer worldwide, and that BCC is a unique type of cancer. Despite the variety of BCC presentations, the majority of them are managed with early intervention. Globally, practice variations may be changing, but the commonality of the disease makes it easier to recognize and treat.
In most cases, surgery is the gold-standard treatment for BCC, but factors such as the size and location of the cancer influence a patient’s treatment path. Age is another consideration because younger patients may not be inclined to choose surgery that can result in scars. No matter the case, early identification is key.
Both dermatologists agreed on the importance of the multidisciplinary tumor boards at their respective institutions. They explained that these boards bring together dermatologists, oncologists, and other surgeons to discuss the best course of action for BCC treatment and that these discussions are particularly helpful considering the increase in advanced BCC cases.
They noted that if surgery is not the immediate choice, Hedgehog inhibitors are effective but come with unwanted adverse effects (AEs), such as hair loss, muscle pains, and loss of taste.
Ibrahim then mentions an alternative BCC treatment—one that’s seen an increased use in dermatology—immune checkpoint inhibitors. Immune checkpoint inhibitors block checkpoint proteins from binding with their partner proteins. This prevents the “off “ signal from being communicated so that the body’s T cells can kill cancer cells. For patients who are unable to tolerate the AEs of Hedgehog inhibitors, cemiplimab-rwlc (Libtayo), a PD-1 immune checkpoint inhibitor, offers hope for an alternative and more tolerable treatment.
“We’re really excited, here, in the field of dermatology, about immune checkpoint therapy, specifically the PD-1 inhibitor path-way, and it’s been exciting to see that for melanoma initially and then [for] Merkel cell carcinoma, squamous cell carcinoma, [and] now basal cell carcinoma,” Patel said. Cemiplimab has prompted dermatologists and oncologists to rethink their approach to locally advanced BCC in patients who have either failed Hedgehog therapy or for whom it is not ideal. Patel noted that the response rate for immune checkpoint inhibitors is in the 20% to 30% range for locally advanced or metastatic BCC.
Ibrahim agreed that the response rates for immune checkpoint inhibitors are encouraging because they are reasonably high from an oncology perspective. Because excision and Hedgehog inhibitors were previously the only viable treatment options for BCC, and they are not always successful, immune checkpoint inhibitors address the current gap in care for BCC.
“Of course, there are safety considerations when we’re using any kind of immuno-therapy and immune checkpoint inhibitors. That’s a whole other conversation that we have to have with our patients,” Ibrahim said. “But for the most part, we know that these medications are very well tolerated. At that point, when the patient has or we as practitioners have felt that the Hedgehog inhibitor’s no longer an ideal treatment for that patient, it’s really nice to have an additional, completely different category of medications and class of agents that we can use.”
Regarding safety considerations for cemiplimab, Patel noted that 90% of patients tolerate the drug well. For the 10% of patients who experience AEs, other medications can be added or the cemiplimab dosing can be adjusted to help reduce AEs.
“Immune checkpoint inhibitors are game changers. They really are the holy grail to oncology,” Patel said. “We’re seeing that across cancer types. We saw that in squamous cell carcinoma in a much higher rate than in basal cell carcinoma. But now we have an option for patients that really had nothing—[just] chemotherapy and radiation, that are really not great for this nonresponsive tumor type. And we’re excited to see what will come down the pipeline with dual combination.”
Patel and Ibrahim agreed that immune checkpoint inhibitors will change the way dermatologists treat BCC. For example, in patients who have a suppressed immune system or have received organ transplants, cancer can wreak havoc on their bodies. Now with immune checkpoint inhibitors like cemiplimab, more options are available for these patients who would, oftentimes, be debilitated by cancer. Immune checkpoint inhibitors will put control back in the hands of dermatologists to effectively and safely treat BCC.
Reference
- Patel V, Sherrif I. New directions in the treatment of basal cell carcinoma. Recognize and Refer. August 22, 2022. Accessed September 12, 2022. https://www.dermatologytimes.com/recognize-and-refer/new-directions-in-the-treatment-of-basal-cell-carcinomaI135%
Articles in this issue
over 3 years ago
A Close Look at Necrobiosis Lipoidicaover 3 years ago
The Dx and Rx of Congenital Malalignment Syndrome in Childrenover 3 years ago
Pediatric Nail Disordersover 3 years ago
Innovation Leads to “Windfall” in Dermatologyover 3 years ago
The Psychosocial Impact of Skin Diseases in Childrenover 3 years ago
Impacts of the Inflation Reduction Act on Doctorsover 3 years ago
A Look at the Rise in Adult Acne in WomenNewsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.