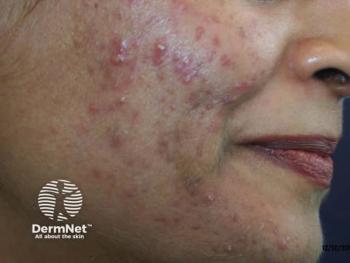
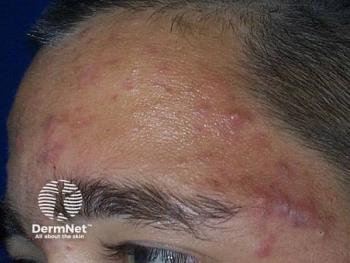
More treatment options emerging for acne, rosacea
In the last three to five years, there has been a tremendous increase in interest in prescription treatments for acne and rosacea, resulting in many promising products entering the marketplace for these conditions.
Thanks to new molecules, new formulations and combinations of old molecules, the number of emerging therapies for acne and rosacea is exploding, says Zoe Diana Draelos, M.D., president of Dermatology Consulting Services, PLLC, and Dermatology Times Medical Editor.
“In the last three to five years, there has been a tremendous increase in interest in prescription treatments for acne and rosacea. That has resulted in many new products entering the marketplace,” she says.
Trifarotene (Aklief, Galderma) was the first new retinoid to earn approval by the U.S. Food and Drug Administration in 20 years. By targeting the retinoic acid receptor gamma, this product offers a more selective mechanism and, therefore, it has a slightly better tolerability than its predecessors.
“It has approval down to age nine, and it can be used for facial and truncal acne,” she says.
MINOCYCLINE
The first topical minocycline preparation (4%) to reach the market is a foam that takes its yellow color from the pigmented minocycline molecule.
“So it’s important to keep it off clothing, towels and pillowcases,” she adds. “The acne lesion count reduction in two parallel 12-week phase 3 trials was significant.”1
The oral antibiotic sarecycline (Seysara, Almirall) is dosed once daily, she notes. “It comes in a variety of different tablet sizes (60 mg, 100 mg and 150 mg), so you can customize dosing for weight. Its indication is for moderate-tosevere facial acne in patients age nine years and older.”
NEW FORMULATIONS
New formulations of existing medications include tazarotene 0.045% lotion, which earned approval by the U.S. Food and Drug Administration in December 2019.
“Unfortunately, tazarotene can be very irritating; so the lotion formulation, with a unique vehicle delivery system, allows tazarotene lotion to be more tolerable and equally as efficacious as tazarotene cream,” Dr. Draelos explains.
Acne treatments under development include besifloxacin (Besivance, Bausch + Lomb), a ciprofloxacin-family antibiotic that is approved to treat bacterial conjunctivitis. A version of this product for facial application completed phase 2 acne studies in late 2019. The results were very promising, so this product will probably move forward, she says.
Spongilla powder offers a new active ingredient, derived from a freshwater sponge, for application as a facial poultice. It is currently in phase 2 clinical trials for the reduction of inflammatory and noninflammatory acne lesions.
“We’re also seeing many clever combination therapies enter the acne market,” Dr. Draelos says.
Examples include benzoyl peroxide with tretinoin, which completed phase 3 trials in 2019, and clindamycin, benzoyl peroxide plus adapalene, now in phase 3.
For patients with
Disclosure:
Dr. Draelos has been an investigator, researcher and/or consultant for Almirall, BioPharmX, Dermata, Foamix, Galderma, Hovione, Ortho Dermatologics, Sol- Gel and Vyome.
Reference:
1. Gold LS, Dhawan S, Weiss J, Draelos ZD, Ellman H, Stuart IA. A novel topical minocycline foam for the treatment of moderate-to-severe acne vulgaris: Results of 2 randomized, double-blind, phase 3 studies. J Am Acad Dermatol. 2019;80(1):168-177.