How Spesolimab Will Revolutionize Generalized Pustular Psoriasis Flares Treatment
Aaron Farberg, MD, presents study data and efficacy of spesolimab for the treatment of generalized pustular psoriasis at the 2022 Fall Clinical Dermatology Conference.
At the 2022 Fall Clinical Dermatology Conference in Las Vegas, Nevada, Aaron Farberg, MD, dermatologist and Mohs surgeon from Dallas, Texas, reviewed the recently FDA-approved spesolimab injection (Spevigo; Boehringer Ingelheim) and its safety and efficacy data for the treatment of generalized pustular psoriasis (GPP).1
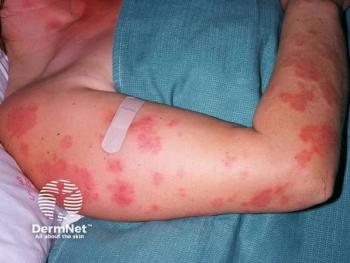
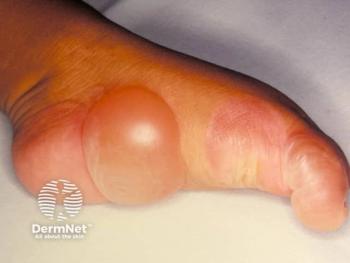
GPP is a rare, severe, relapsing disease that is not multisystemic and can appear on many different areas of the body. It’s important to distinguish that GPP is different from plaque psoriasis. Plaque psoriasis is an adaptive immune response, it’s an autoimmune disease, and it’s driven by the interleukin (IL) 23 pathway. GPP is innate immune inflammation, neutrophilic, it’s driven by the IL-36 pathway, and it can also be associated with mutations in IL36RN in some patients. GPP flares can also occur with or without systemic inflammation, and with or without concomitant plaque psoriasis.
There are many different factors that can trigger GPP flares such as:
- Overexposure to the sun
- Upper respiratory tract infections
- Sudden withdrawal of steroids and exposure to certain medications
- Hormonal changes
- Pregnancy
- Stress
Patients with GPP typically present as sick, anorexic, feverish, or anxious. They can also have life-threatening complications, multi-system type failure issues with their heart and kidneys, and sepsis infections, often requiring emergency care.
The IL-36 signaling pathway is a member of the IL-1 superfamily and includes 3 agonists, IL-36α, IL-36β, and IL-36γ, and includes 1 antagonist, IL-36RA. IL-36 agonists bind to the IL-36 receptors (and IL-1 RAcP) and activate MyD88, MAPK, and NF-κB signaling pathways to create an inflammatory response.
“We don't have any approved [GPP] treatments, until most recently was Spevigo. Also importantly, none of those other agents actually worked on the IL-36 pathway specifically. We know now how important it is to find various specific treatments for these types of diseases,” said Farberg.
Spesolimab is the first FDA-approved IL-36 receptor antagonist indicated for the treatment of adults with GPP flares. Spesolimab is contraindicated in patients with severe or life-threatening hypersensitivity to spesolimab. Some reactions have included drug reaction with eosinophilia and systematic symptoms (DRESS). One of the most common adverse effects to watch out for is infections. It’s important to confirm before treatment with spesolimab that patients do not have any outstanding infections.
Farberg then reviewed data from the recent spesolimab clinical trial, Effisayil –1 (NCT03782792). Out of 53 participants, 43% (n=35) achieved a Generalized Pustular Psoriasis Physician Global Assessment (GPPPGA) total score of 0 or 1 at week 1. The GPPPGA total score is determined by average subscores of pustules, scaling, and erythema. Next, Farberg demonstrated how spesolimab’s efficacy was sustained for up to 12 weeks of the study duration. After 12 weeks, many patients continued to show rapid GPP flare improvement.
Also part of the Effisayil –1 trial, patients were evaluated using the pain Visual Analogue Scale (VAS) and the Dermatology Life Quality Index (DLQI). From baseline through week 12, patients reported a dramatic improvement in the pain VAS and DLQI.
“We looked at DLQI as well as the pain Visual Analog Scale. How are they feeling? Is [spesolimab] improving how they feel? It's good to see these graphs go down. And obviously, you can see they go down, they go down quickly, and they stay down,” said Farberg.
The key takeaways from Farberg’s presentation were:
- GPP is a life-threatening, debilitating disease, recognized by small, sterile pustules on the arms, legs, and trunk
- GPP and plaque psoriasis are 2 different diseases based on clinical findings, genetics, pathophysiology, and severity
- GPP is driven by dysfunctional IL-36 signaling, creating uncontrolled inflammation
- Spesolimab was shown to be effective in improving affected skin of adults with GPP flares
- The most common adverse events (<5%) in Effisayil –1 were asthenia, fatigue, nausea, vomiting, headache, pruritis and prurigo, infusion site hematoma, and urinary tract infection
Dermatologists can find more information on
Reference
- Farberg A. Spevigo: A breakthrough treatment for patients with generalized pustular psoriasis. 2022 Fall Clinical Dermatology Conference. October 20, 2022. Las Vegas, Nevada.
Newsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.