- Dermatology Times, December 2022 (Vol. 43. No. 12)
- Volume 43
- Issue 12
Erythropoietic Protoporphyria: A Brief Review
Erythropoietic protoporphyria (EPP) is a life-long and debilitating disorder resulting from defects in the enzymes in heme synthesis. Patients with the disease are cast indoors due to extreme photosensitivity.
Background
Erythropoietic protoporphyria (EPP) is a disorder in one of two enzymes in the heme synthesis pathway. A deficiency of the enzyme ferrochelatase (FECH) or a gain-of-function mutation in aminolevulinic acid synthase (ALAS2) are two common mutations seen in this disease. The result of these aberrancies is an accumulation of protoporphyrin in red blood cells which leads to a non-blistering, photosensitive, and painful cutaneous presentation. Over time, repeated insults to the skin with exposure to sunlight results in pebbly thickening of the skin and wax-like scarring of the face. In a subset of patients with EPP, liver disease such as cirrhosis may also develop.1
Epidemiology
EPP is the third most common porphyria in the world. With an incidence of 2-5 per 1 million, this disease affects 1 in 74,300 individuals both male and female around the world.2 Onset typically occurs during childhood with the average age of diagnosis around 4 years;3 however, adolescent and adult onset could also occur.
Clinical Presentation
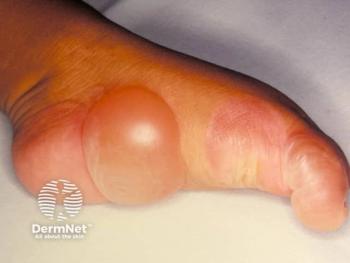
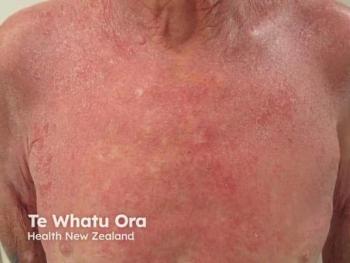
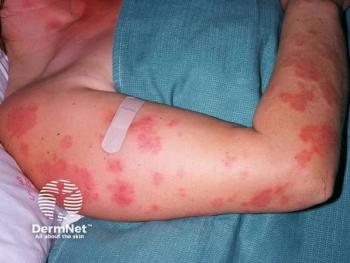
Patients with EPP primarily suffer from extreme photosensitivity following brief and limited sun exposure. The protoporphyrin that has accumulated in their red blood cells and deposited in dermal blood vessels are photoactivated and result in intense skin burning, erythema, and pruritus within hours of sun exposure.4 Common locations of dermal findings include frequently sun-exposed areas such as the nose, cheeks, and dorsum of the hands. Severity of dermatologic findings are related to the duration of sun exposure. As expected, due to the avoidance of sunlight, patients with EPP are more likely to be vitamin D deficient and have increased risk for osteoporosis.1
Extracutaneous sequela such as liver damage is a rare but severe complication for patients with EPP. The pathogenesis of this complication is potentially secondary to the cholestatic and hepatotoxic effects of protoporphyrin which accumulates in the liver.4 For this reason, patients occasionally present with rapidly progressive liver disease including fatal hepatic failure or severe chronic cirrhosis which further precipitates the protoporphyrin accumulation in a vicious cycle. A more common complication of EPP is pigmented gallstones. Protoporphyrin is lipid-soluble and may crystallize in bile. In tandem with cholestatic changes in the liver, these patients are at increased risk for gallstones at a young age.1,4
Diagnostics
Due to the early presentation of this disease, clinical evaluation during infancy allows for appropriate recommendations to be made. Given that protoporphyrin is not water soluble, urinalysis is generally negative in these patients. Instead, samples of blood are evaluated for red blood cells containing an increased amount of total and free protoporphyrin. The distinction of free protoporphyrin is important to rule out other causes of porphyria such as iron or lead toxicity which presents with a zinc compounded porphyria.4 Five to ten times greater than the upper limit of normal serum protoporphyrin (80mcg/dL) is diagnostic of the disease.1 Autosomal dominant transmission is most common, so genetic testing or gene sequencing could then be pursued to determine the specific enzyme affected.
With liver damage being a severe complication of the disease, it is important to obtain liver function tests at the time of diagnosis to establish a baseline and monitor throughout the disease course.1,4 If necessary, liver biopsies could be performed to assess the severity of the liver damage and whether the patient will require liver transplantation.3,4 Even with pursuit of a liver transplantation, the underlying cause is not being addressed and the hepatotoxic effects of the accumulation of protoporphyrin may still continue.
Treatment Options
There is currently no effective way to decrease protoporphyrin levels in the body in patients with EPP. The mainstay of treatment is prevention of symptoms. Patients with EPP are advised to avoid sunlight, wear protective clothing, apply sunscreen, remain in shaded areas if outside, and use light filters within their homes. However, such drastic behaviors are difficult to implement and have a grave impact on the patient’s quality of life. Secondary to lack of sun exposure, EPP patients tend to present with vitamin D deficiency and increased risk of osteoporosis. Thus, supplements such as vitamin D and calcium are helpful for counteracting these effects.1
Beta-carotene has been noted to provide EPP patients with mild increased tolerance to sunlight, although its efficacy is skeptical amongst experts in the field.5 In addition, higher doses of beta-carotene can lead to yellow skin pigmentation and higher risk of lung cancer among current smokers, making this a less desirable treatment option for patients.6
In 2019, afamelanotide (a synthetic analog of alpha melanocyte stimulating hormone or MSH) gained FDA approval in the United States for the treatment of EPP.7 Afamelanotide acts by binding to the melanocortin 1 receptor in dermal cells and producing eumelanin, a photoprotective molecule.8 In addition, there are antioxidant activities, increased melanin synthesis, and inflammation modulation with afamelanotide.7 This medication has been shown to improve patient’s quality of life and has great compliance rates,9 when compared to beta-carotene. Further, afamelanotide has minimal side effects with low patient discontinuation rates.9 Reports of nausea, headache, and temporary skin darkening have been noted with use of this medication.8 Overall, afamelanotide improves patient tolerance to sunlight and has a mild adverse side effect profile making it a desirable treatment option for patients with EPP. Despite its success, this medication only provides temporary relief and improvement in quality of life without addressing the underlying pathology. To date, there is no effective treatment for decreasing the accumulation of porphyrins which serves as the main culprit of disease.
Discussion
Patients with erythropoietic protoporphyria suffer from a disease that prevents them from exposing their skin to sunlight whether outdoors or within their home. This debilitating disease has no current cure and treatments are focused on symptomatic control and prevention. Availability of treatments such as afamelanotide is a crucial milestone that has been reached for these patients and should be advocated for in all patients with EPP suffering from extreme photosensitivity.
References
1. Ahmed JN, Masood S. Erythropoietic Protoporphyria. In: StatPearls [Internet]. May 08, 2022 2022;
2. APF. Erythropoietic Protoporphyria (EPP) and X-Linked Porphyria (XLP). American Porphyria Foundation (APF). Accessed October 14, 2022.
3. NORD. Erythropoietic Protoporphyria and X-Linked Protoporphyria. National Organization for Rare Disorders (NORD). Accessed October 14, 2022.
4. Thapar M, Bonkovsky HL. The diagnosis and management of erythropoietic protoporphyria. Gastroenterol Hepatol (N Y). Aug 2008;4(8):561-6.
5. Minder EI, Schneider-Yin X, Steurer J, Bachmann LM. A systematic review of treatment options for dermal photosensitivity in erythropoietic protoporphyria. Cell Mol Biol (Noisy-le-grand). Feb 16 2009;55(1):84-97.
6. Köpcke W, Krutmann J. Protection from sunburn with beta-Carotene--a meta-analysis. Photochem Photobiol. Mar-Apr 2008;84(2):284-8. doi:10.1111/j.1751-1097.2007.00253.x
7. Heymann W. For patients with erythropoietic protoporphyria, the tide has come in. Dermatology World Insights and Inquiries. May 12, 2021 2021;3(19)
8. Langendonk JG, Balwani M, Anderson KE, et al. Afamelanotide for Erythropoietic Protoporphyria. N Engl J Med. Jul 2 2015;373(1):48-59. doi:10.1056/NEJMoa1411481
9. Biolcati G, Marchesini E, Sorge F, Barbieri L, Schneider-Yin X, Minder EI. Long-term observational study of afamelanotide in 115 patients with erythropoietic protoporphyria. Br J Dermatol. Jun 2015;172(6):1601-1612. doi:10.1111/bjd.13598
Articles in this issue
about 3 years ago
Cost-Effectiveness of Treating Advanced Melanomaabout 3 years ago
Q&A: Muneeb Shah, DO, Discusses What it Means to Have Healthy Skinabout 3 years ago
2022 Top Trends and What’s Next in Dermatologyabout 3 years ago
Pulsed Dye Lasers for Cosmetic Proceduresabout 3 years ago
Top Meeting Takeaways From the Fall Clinical Conferenceabout 3 years ago
Was It Marketing or Was It Misrepresentation?about 3 years ago
Treatment of Plaque Psoriasis in Childrenabout 3 years ago
A Conversation About JAK Inhibitors in Atopic Dermatitisabout 3 years ago
Holistic Treatment for the Management of Atopic DermatitisNewsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.