- Dermatology Times, June 2022 (Vol. 43. No. 6)
- Volume 43
- Issue 6
- Pages: 34
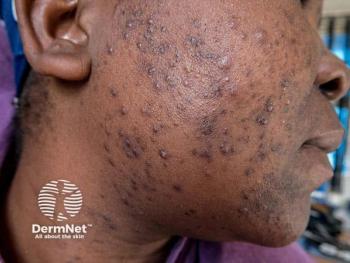
Acne Update: 2022’s Top 5 Trends
Fresh spins on established treatments and newly FDA-approved drugs and devices provide a framework for changing therapies and modalities for acne regimens.
Acne treatment has advanced by leaps and bounds over the past several years, with breakthrough advances and fresh spins on classic treatments. All of this can make it hard to determine which rethought standard treatments and new therapies should be a regular part of every dermatologist’s treatment toolbox in 2022.
1. Addressing Antibiotics
Topical minocycline was born from the need for an effective topical antibiotic with minimal antibiotic resistance. Enter minocycline foam 4% (Amzeeq; Journey Medical Corporation), which made topical tetracycline antibiotic delivery possible with a uniquely micronized formulation in a moisturizing vehicle.1 Topical minocycline was first approved by the FDA in October 2019. One distinct advantage of topical vs oral delivery of minocycline is limited systemic absorption. Even after 21 days of daily use, serum levels of minocycline are still more than 700-fold lower than after exposure to 1 dose of oral minocycline.2
Although oral minocycline has somewhat fallen out of favor in recent times due to rare but serious adverse events such as hypersensitivity reactions, drug-induced hyperpigmentation, and photosensitivity, none of these were reported with topical minocycline.3,4 Another distinct advantage is potential simplification of therapy and increased tolerability.
Traditional topical antibiotic acne therapy relies on a combination approach, often with an antimicrobial like benzoyl peroxide to combat bacterial resistance, which can subsequently lead to irritation in some patients. In vitro studies show that topical minocycline can be effectively delivered at concentrations that may not drive antibiotic resistance to Cutibacterium acnes—in contrast totraditional first-line topical antibiotics like clindamycin and erythromycin—and thus obviate a need for benzoyl peroxide.5
Keep an eye on further evolution in the topical antibiotic space, and watch for additional research. In March 2022, Chinese researchers published data on a new metric they developed called the C acnes quantitative polymerase chain reaction–based antibiotics resistance assay (ACQUIRE) “to enable fast and accurate detection of C acnes macrolide resistance in clinical settings, representing an opportunity to administer antibiotics more wisely and improve the quality of care.” Based on results of the cross-sectional observational study (n=915), the authors concluded, “The macrolide resistance of C acnes is much higher than previously reported. Integrating ACQUIRE into acne treatment modalities may eliminate macrolide misuse and achieve better clinical improvements.”6
2. Suppressing Sebum
Clascoterone is an anti-androgen and competitive antagonist of dihydrotestosterone (DHT) for the androgen receptor. By blocking binding of DHT to the androgen receptor, clascoterone inhibits production of sebum, pro-inflammatory cytokines, and inflammatory follicular activity.7
Clascoterone cream 1% (Winlevi; Sun Pharmaceutical Industries, Inc), which was approved by the FDA in August 2020 and became available in the US in November 2021, targets sebum production driving inflammation and overgrowth of C acnes. The first novel acne molecule in decades, clascoterone cream 1% showed significant improvement in acne on the face and trunk in clinical trials.8,9
Although similar in molecular structure to spironolactone, clascoterone has minimal systemic antiandrogen effects, making it an enticing topical alternative to oral spironolactone, which has limited use in women who may be pregnant or breastfeeding as well as men in general. Look for emerging real-world data about its use as monotherapy as well as in combination with topical retinoids and/or antimicrobials.
3. Reinventing Retinoids
Topical retinoids are foundational for successful topical acne treatment but problematic for patients whose challenges may include tolerability and complexity of regimens. Two established retinoids that have found new vehicles are tretinoin lotion 0.05% (Altreno; Ortho Dermatologics) and tazarotene lotion 0.045% (Arazlo; Ortho Dermatologics), approved by the FDA in August 2018 and December 2019, respectively. Clinical trials for both formulations demonstrated very favorable tolerability, which has been an issue with conventional retinoid formulations in the past.10,11
Look for vehicle innovation to become as crucial as drug discovery with the next generation of topical therapies—especially when it comes to encouraging regular medication use in chronic inflammatory skin conditions. Striking the balance of efficacy and tolerability is especially important when treating skin of color (SOC) patients who are more prone to develop dyspigmentation with treatment-related irritant contact dermatitis.
Trifarotene cream 0.005% (Aklief; Galderma Laboratories, LP), approved by the FDA in October 2019, is the first and only retinoic acid receptor (RAR) agonist with the advantage of increased specificity for the RARγ receptor, the predominant isoform in the skin. This increased specificity lends to its use over a wide body surface area (eg, trunk), which was previously a limitation with traditional topical retinoids.12 In addition to clinical trial data showing the cream’s efficacy on both the face and body, another study, LEAP (NCT05089708),is underway with an end point of postinflammatory hyperpigmentation—a major acne-related concern for patients with SOC.13
Another recent study notes that more research is needed into emerging and established agents to create optimal treatment plans that also take cost-effectiveness into consideration.14
4. Combining Classics
Advancements in topical formulation have also enabled new fixed-combination acne treatments such as tretinoin 0.1%/benzoyl peroxide 3% cream (Twyneo; Galderma Laboratories, LP), which the FDA approved in July 2021. Previous studies have shown that traditional formulations of tretinoin are unstable in the presence of both benzoyl peroxide and sunlight, leading to cumbersome treatment regimens when use of both products is desired.14,15 In this new formulation, both ingredients are microencapsulated in silica core shells, giving them stability and enabling gradual delivery into the skin.16
Another exciting combination is clindamycin phosphate 1.2%/benzoyl peroxide 3.1%/adapalene 0.15% gel (IDP-126), which is currently under investigation. Phase 2 clinical trial results showed additional synergy for this triple combination compared with conventional dual and monotherapy—in addition to the obvious advantages of a simplified treatment regimen.17
In addition, Stubborn Blackheads Daily Acne Facial Serum (Johnson&Johnson), a novel 10% multiacid serum containing exfoliating 5% glycolic acid, 2.5% mandelic acid (a lipophilic hydroxy acid) 2.5% gluconolactone (a polyhydroxy acid with humectant and skin-clarifying prop- erties), and 0.5% salicylic acid, showed significant reduction in inflammatory and noninflammatory lesions in patients with mild to moderate acne.18
5. Emerging Energy
Energy-based devices have become a critical part of the dermatology armamentarium. Selective photothermolysis of sebaceous glands with a 1726-nm Raman fiber laser appears to be an efficacious approach toward acne treatment, allowing for selective destruction of sebaceous glands for rapid and durable acne control with few treatments and minimal collateral damage.19,20,21
Adding to the laser’s safety, melanin does not meaningfully absorb energy at the 1726-nm wavelength, suggesting that this approach may have broader applicability across a variety of skin pigmentation. Photodynamic therapy (with 5-aminolevulinic acid) has been one of the most widely published light-based treatments for acne during the past several years. Numerous protocols have been suggested, with each adjusting for incubation time and penetration depth with red light, blue light, and even ambient daylight to target sebaceous glands and/or achieve antimicrobial effects.22,23,24
In March 2022, the FDA gave the first 510(k) clearance to an energy-based device for treating mild, moderate, and severe acne (AviClear; Cutera, Inc).25 Although official recommendations for photodynamic therapy are limited by heterogeneous data and lack of well-controlled, standardized trials, look for additional investigations into laser and light-based therapies over the next several years as these adjunctive approaches become more widespread.
References
1. Kircik L, Del Rosso JQ, Weiss JS, et al. Formulation and profile of FMX101 4% minocycline topical foam for the treatment of acne vulgaris. J Clin Aesthet Dermatol. 2020;13(4):14-21.
2. Jones TM, Ellman H, deVries T. Pharmacokinetic comparison of once-daily topical minocycline foam 4% vs oral minocycline for moderate-to-severe acne. J Drugs Dermatol. 2017;16(10):1022-1028.
3. Gold LS, Dhawan S, Weiss J, Draelos ZD, Ellman H, Stuart IA. A novel topical minocycline foam for the treatment of moderate-to-severe acne vulgaris: results of 2 randomized, double-blind, phase 3 studies. J Am Acad Dermatol. 2019;80(1):168-177. doi:10.1016/j.jaad.2018.08.020
4. Raoof TJ, Hooper D, Moore A, et al. Efficacy and safety of a novel topical minocycline foam for the treatment of moderate to severe acne vulgaris: a phase 3 study. J Am Acad Dermatol. 2020;82(4):832-837. doi:10.1016/j.jaad.2019.05.078
5. Sutcliffe J, McLaughlin R, Webster G, et al. Susceptibility of Cutibacterium acnes to topical minocycline foam. Anaerobe. 2020;62:102169. doi:10.1016/j.anaerobe.2020.102169
6. Rosette C, Agan FJ, Mazzetti A, Moro L, Gerloni M. Cortexolone 17α-propionate (clascoterone) is a novel androgen receptor antagonist that inhibits production of lipids and inflammatory cytokines from sebocytes in vitro. J Drugs Dermatol. 2019;18(5):412-418.
7. Hebert A, Thiboutot D, Stein Gold L, et al. Efficacy and safety of topical clascoterone cream, 1%, for treatment in patients with facial acne: two phase 3 randomized clinical trials. JAMA Dermatol. 2020;156(6):621-630. doi:10.1001/jamadermatol.2020.0465
8. Eichenfield L, Hebert A, Gold LS, et al. Open-label, long-term extension study to evaluate the safety of clascoterone (CB-03-01) cream, 1% twice daily, in patients with acne vulgaris. J Am Acad Dermatol. 2020;83(2):477-485. doi:10.1016/j.jaad.2020.04.087
9. Tanghetti EA, Kircik LH, Green LJ, et al. A phase 2, multicenter, double-blind, randomized, vehicle-controlled clinical study to compare the safety and efficacy of a novel tazarotene 0.045% lotion and tazarotene 0.1% cream in the treatment of moderate-to-severe acne vulgaris. J Drugs Dermatol. 2019;18(6):542.
10. Tyring SK, Kircik LH, Pariser DM, Guenin E, Bhatt V, Pillai R. Novel tretinoin 0.05% lotion for the once-daily treatment of moderate-to-severe acne vulgaris: assessment of efficacy and safety in patients aged 9 years and older. J Drugs Dermatol. 2018;17(10):1084-1091.
11.Tan J, Thiboutot D, Popp G, et al. Randomized phase 3 evaluation of trifarotene 50 μg/g cream treatment of moderate facial and truncal acne. J Am Acad Dermatol. 2019;80(6):1691-1699. doi:10.1016/j.jaad.2019.02.044
12. Drake L, Reyes-Hadsall S, Barbieri JS, Mostaghimi A. New developments in topical acne therapy. Am J Clin Dermatol. 2022;23(2):125-136. doi:10.1007/s40257-021-00666-9
13. AkLief evaluation in acne-induced post-inflammatory hyperpigmentation (LEAP). ClinicalTrials.gov. Updated February 1, 2022. Accessed May 9, 2022. https://clinicaltrials.gov/ct2/show/NCT05089708
14. Martin B, Meunier C, Montels D, Watts O. Chemical stability of adapalene and tretinoin when combined with benzoyl peroxide in presence and in absence of visible light and ultraviolet radiation. Br J Dermatol. 1998;139(suppl 52):8-11. doi:10.1046/j.1365-2133.1998.1390s2008.x
15. Nyirady J, Lucas C, Yusuf M, Mignone P, Wisniewski S. The stability of tretinoin in tretinoin gel microsphere 0.1%. Cutis. 2002;70(5):295-298.
16. Del Rosso J, Levy-Hacham O, Mizrahi O. Efficacy and safety of microencapsulated benzoyl peroxide 3% and microencapsulated tretinoin 0.1% (E-Bpo/E-Atra) in acne vulgaris: results from two randomized controlled clinical trials. SKIN J Cutan Med. 2021;5(1). doi:
17. Stein Gold L, Baldwin H, Kircik LH, et al. Efficacy and safety of a fixed-dose clindamycin phosphate 1.2%, benzoyl peroxide 3.1%, and adapalene 0.15% gel for moderate-to-severe acne: a randomized phase II study of the first triple-combination drug. Am J Clin Dermatol. 2022;23(1):93-104. doi:10.1007/s40257-021-00650-3
18. Kosmoski G, Smith H, Miller D.Novel 10% multiacid serum provides clinical acne and complexion benefits in subjects with mild to moderate acne. Poster presented at: 2022 American Academy of Dermatology Annual Meeting; March 25-29, 2022; Boston, MA.
19. Sakamoto FH, Doukas AG, Farinelli WA, et al. Selective photothermolysis to target sebaceous glands: theoretical estimation of parameters and preliminary results using a free electron laser. Lasers Surg Med. 2012;44(2):175-183. doi:10.1002/lsm.21132
20. Anderson RR, Farinelli W, Laubach H, et al. Selective photothermolysis of lipid-rich tissues: a free electron laser study. Lasers Surg Med. 2006;38(10):913-919. doi:10.1002/lsm.20393
21. Tanghetti E, Geronemus R, Bloom B, Anderson RR, Ross EV, Sakamoto FW. Safety and efficacy data in a pilot study of the treatment of acne with a fiber laser. Presented at: 40th American Society for Laser Medicine and Surgery Annual Conference; April 29-May 3, 2020; Phoenix, AZ.
22. Sakamoto FH, Torezan L, Anderson RR. Photodynamic therapy for acne vulgaris: a critical review from basics to clinical practice: part II. understanding parameters for acne treatment with photodynamic therapy. J Am Acad Dermatol. 2010;63(2):195-211; quiz 211-212. doi:10.1016/j.jaad.2009.09.057
23. Sakamoto FH, Lopes JD, Anderson RR. Photodynamic therapy for acne vulgaris: a critical review from basics to clinical practice: part I. acne vulgaris: when and why consider photodynamic therapy? J Am Acad Dermatol. 2010;63(2):183-193; quiz 193-194. doi:10.1016/j.jaad.2009.09.056
24. Hongcharu W, Taylor CR, Chang Y, Aghassi D, Suthamjariya K, Anderson RR. Topical ALA-photodynamic therapy for the treatment of acne vulgaris. J Invest Dermatol. 2000;115(2):183-192. doi:10.1046/j.1523-1747.2000.00046.x
25. FDA clears Cutera’s AviClear acne device. News release. Cutera Inc. March 25, 2022. Accessed May 11, 2022. https://www.businesswire.com/news/home/20220317006022/en/FDA-Clears-Cutera%E2%80%99s-AviClear%E2%84%A2-Acne-Device
Articles in this issue
over 3 years ago
Hidradenitis Suppurativa in Pregnancyover 3 years ago
Explore New Tactics for Acne Scarsover 3 years ago
Meeting the Challenges of Acne in Adultsover 3 years ago
Protect Yourself From Risk With Strategies Beyond Insuranceover 3 years ago
Hair Happeningsover 3 years ago
Think Globally to Combat Acneover 3 years ago
Targeted Solutions Simplify Widespread Itch Carealmost 4 years ago
Epsolay Receives FDA ApprovalNewsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.