- General Dermatology
- Eczema
- Alopecia
- Aesthetics
- Vitiligo
- COVID-19
- Actinic Keratosis
- Precision Medicine and Biologics
- Rare Disease
- Wound Care
- Rosacea
- Psoriasis
- Psoriatic Arthritis
- Atopic Dermatitis
- Melasma
- NP and PA
- Skin Cancer
- Hidradenitis Suppurativa
- Drug Watch
- Pigmentary Disorders
- Acne
- Pediatric Dermatology
- Practice Management
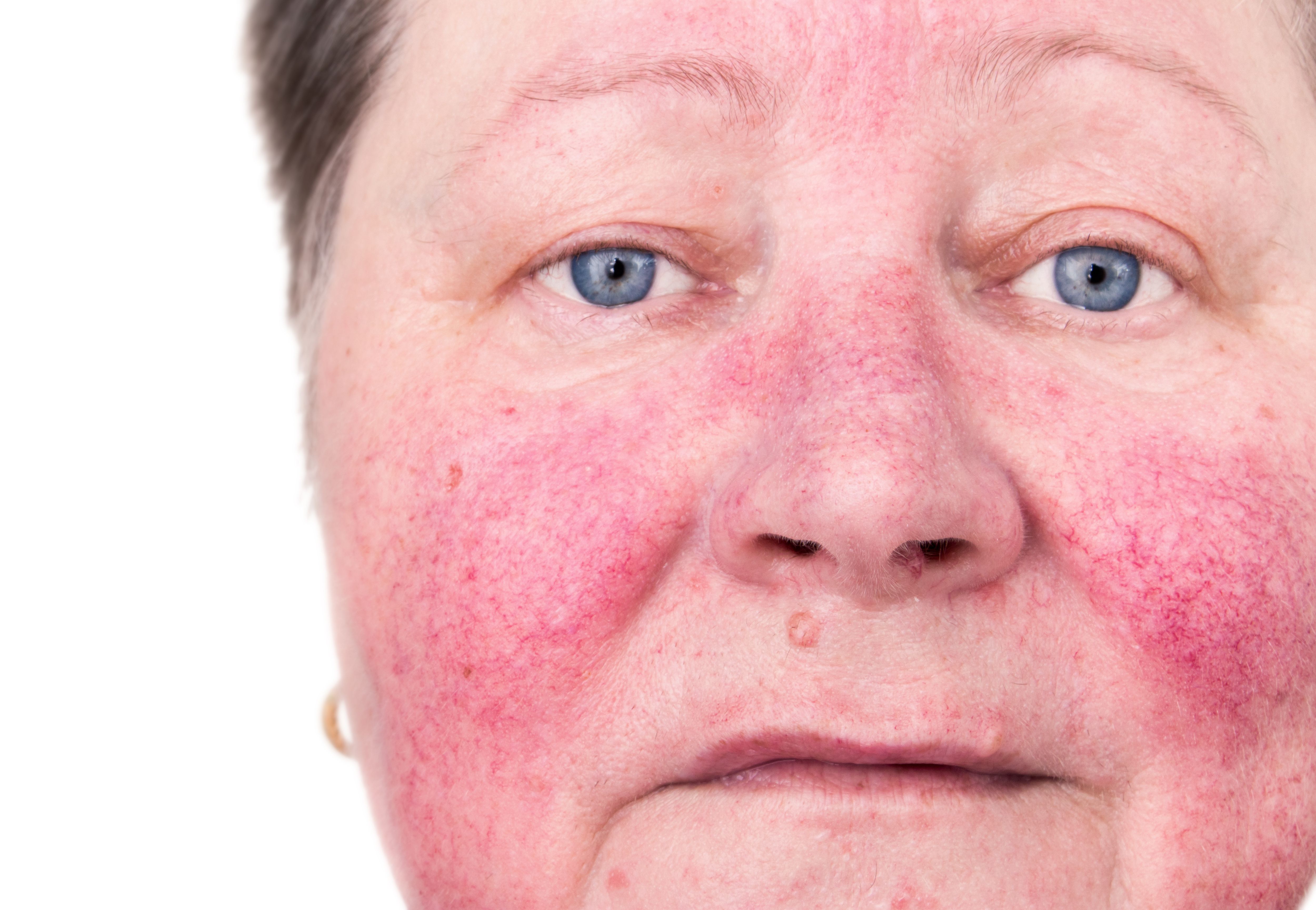
Prolonged Remission in Rosacea: A Breakthrough With Subantibiotic Dose of Oral Doxycycline
Researchers found that continued use of once-daily subantibiotic dose oral doxycycline 40 mg modified-release capsules significantly reduced the relapse rate and inflammatory lesion counts in patients with moderate-to-severe rosacea.
Achieving and sustaining remission in rosacea patients presents a significant challenge, given the cyclical nature of exacerbations and remissions.
“Treatment combining topical metronidazole gel 1% and subantibiotic dose doxycycline (SDD) [dosing to avoid antibiotic resistance, previously referred to as subantimicrobial], administered as once-daily 40 mg modified-release capsules [30 mg of immediate-release and 10 mg delayed-release via beads], has been shown to produce a more rapid and greater reduction of papules and pustules compared with topical metronidazole monotherapy,” explained James Del Rosso, DO; Sam Brantman, PharmD; and Hilary Baldwin, MD, in their recent research.1
However, the long-term efficacy of SDD40 in sustaining remission remained unexplored. Investigators launched a study to evaluate that and presented data in a poster session at Maui Derm NP+PA Fall 2023 in Asheville, North Carolina from September 27-30.1
Study Design
This comprehensive 2-part study aimed to evaluate the extended remission capabilities of SDD40 monotherapy. Part 1 involved a multicenter, 12-week open-label study wherein adults with moderate or severe inflammatory lesions of rosacea received SDD40 in combination with topical metronidazole gel 1%. Part 2 was a 40-week randomized, double-blind, placebo-controlled trial. Successfully treated subjects were randomly assigned to receive once-daily SDD40 or placebo capsules. The primary objective was to assess relapse rates and efficacy during long-term SDD40 use versus placebo.
Study Results
Part 1 enrolled 235 subjects, of which 65 in both the SDD40 treatment group and placebo group met the criteria for treatment success at week 12 and proceeded to part 2. At the end of part 2, a remarkable outcome emerged: only 13.8% of subjects treated with SDD40 experienced relapse, compared to 27.7% in the placebo group (P < .05). Furthermore, there were significant differences in the median change in inflammatory lesion counts (P < .05). Adverse events were generally mild or moderate, with most being unrelated to the treatment. Notably, subjects treated with SDD40 reported greater improvement in stinging and burning sensations.
Conclusions and Take Home Pearls
This study reveals that after an initial 12-week treatment regimen, continued use of once-daily SDD40 significantly reduces the relapse rate and inflammatory lesion counts in subjects with moderate-to-severe rosacea. These findings herald a major advancement in rosacea management, offering new hope for sustained remission and improved quality of life for patients.
Reference
1. Del Rosso J, Brantman S, Baldwin H. Maximizing remission in rosacea with subantibiotic dose oral doxycycline 40 mg modified-release capsules once daily. Poster presented at: Maui Derm NP+PA Fall 2023; September 27-30, 2023; Asheville, NC.