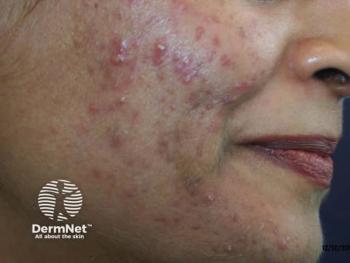
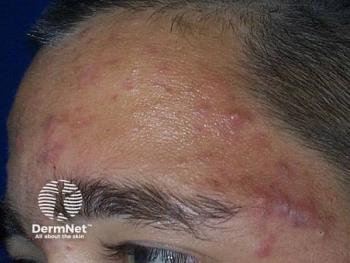
Masculinizing Hormone Therapy Linked to Acne Diagnosis
A cohort study investigated the association between MHT and acne diagnosis.
In a study published by JAMA Dermatology, examined why there may be an association between masculinizing hormone therapy (MHT) and acne.1
The objective of this analysis was to evaluate the risk of acne in a large population of transgender patients that received MHT and the clinical risk factors for acne diagnosis.
“The importance of mitigating health disparities in gender diverse individuals is becoming increasingly recognized as this population seeks access to standards of excellence in healthcare,” said Klint Peebles, MD, FAAD, board certified dermatologist with the Kaiser Permanente, Mid-Atlantic Permanente Medical Group in Washington D.C., United States. Peebles was one of the study researchers. “It is well-established that lack of access to gender-affirming healthcare is a risk factor for poor mental health outcomes, including suicide, as well as harm to well-being and quality of life.”
The retrospective cohort study contained 988 patients who started MHT treatment between January 1, 2014 and December 31, 2017. They also had at least 1 year of follow up from treatment. The data was analyzed between 2 weeks starting on September 1, 2019 and ending on September 15, 2019.
Population data was comprised of patients receiving MHT who were 18 years or older at the start of treatment and were assigned female sex at birth. Data for this study was attained by using electronic health records from a community health center serving the sexual and gender minority community.
The primary endpoint for acne was defined by the International Statistical Classification of Diseases, Tenth Revision, Clinical Modification codes. The study then investigated the prevalence and incidence rates (IRs) of acne over 2 years after initial MHT.
“We know that acne is a common condition among gender diverse persons receiving masculinizing hormone therapy [MHT], but large-scale studies of risk factors and treatment outcomes are lacking, particularly given that gender identity demographic variables are rarely routinely integrated into acne research,” Peebles said.
After the data was collected, a series of univariate analyses for all factors was calculated for association with acne diagnosis. It was then followed by multivariate analyses to test for independent predictors.
The median age of the 988 patients was 25.8 years with an interquartile range of 20.8-28.2 years. Overall, there was acne prevalence of 31.1% among patients. One year after therapy, the incident rate was 19% and the 2-year rate increased by 4% totaling to 25.1%. After looking at the ages of the patients and the prevalence of acne, it was found that the younger the patient was when they began therapy, the higher likelihood of developing acne. The median age for those who developed acne was 22.4 years versus 24.7 years among those who did not.
“Prior studies were on smaller cohorts, but these found much higher prevalence of acne than the current study,” said Richard Gallo, MD, PhD, a professor and founding chairman of the department of dermatology at the University of California, San Diego, California.
Peebles explains why the higher prevalence rate was found in other studies compared to this one.
“Prior studies demonstrated even higher prevalence rates though these are limited by much smaller sample sizes and methodological differences that could have led to the detection of milder acne cases,” Peebles explained.
Gallo also noted how, “Younger patients had the highest incidence but the lowest Testosterone.” Peebles explained why this might be.
“Our study interestingly found a greater likelihood of acne development in younger individuals. Additionally, serum testosterone levels were lowest in this group, further corroborating prior assertions that acne prevalence is likely related to factors other than serum levels. However, further studies are warranted to determine the impact of these other factors, including but not limited to testosterone formulation, routes of administration, and dosing,” Peebles said.
Gallo commented on what this means for dermatologists, “They should recognize and consul on increased risk, consider the age of the patient, and reaffirm that acne is a complex disorder that cannot be simply predictable based on MHT alone.”
Acne is a common condition among transgender patients on MHT. The study found that the prevalence increased from 6.3% to 31.1% following MHT initiation. Patients aged 18 to 21 years appear to be the most likely to develop acne after MHT initiation.
“More robust dedicated research is needed to develop evidence-based guidelines for the management of acne in gender diverse individuals. In the meantime, dermatologists should be aware of the high acne prevalence in persons undergoing treatment with MHT and should emphasize the importance of the routine integration of the dermatology specialty in gender-affirming care,” Peebles said. “Acne research, whether focusing specifically on gender minority individuals or not, should integrate gender identity into the study demographic variable collection, which also should become standardized more broadly across scientific research.”
“Future prospective studies and investigations to determine optimal therapy for these patients is still needed,” Gallo agreed.
Disclosures:
Peebles and Gallo report no relevant disclosures.
References:
1. Thoreson N, Park JA, Grasso C, et al. Incidence and factors associated with acne among transgender patients receiving masculinizing hormone therapy. JAMA Dermatology. 2021;157(3):290-295. doi:10.1001/jamadermatol.2020.5347