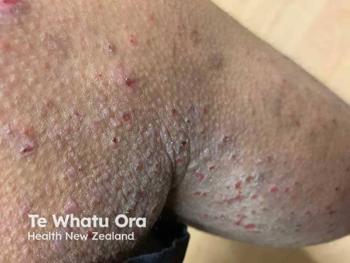
Dupilumab Demonstrates Long-Term Safety for Moderate to Severe AD
The fully human monoclonal antibody is currently approved in more than 60 countries.
The European Medicines Agency will update the dupilumab (Dupixent, Regeneron Pharmaceuticals, Inc) summary of product characteristics with long-term safety results for adults with moderate-to-severe atopic dermatitis based on positive feedback by the Committee for Medicinal Products for Human Use.
Looking at data from a single-arm, phase 3 open-label extension (OLE) trial, the long-term safety profile in adults with atopic dermatitis observed up to 3 years was generally consistent with what was observed in the controlled phase 3 trials. Further, the trial evaluated the long-term safety of dupilumab 300 mg weekly in adults who had previously participated in controlled dupilumab trials or had been screened for a phase 3 trial. The dupilumab dose that is approved in adults is 300 mg every other week, according to a Regeneron press release.
Dupilumab is the only biologic approved in the European Union for children as young as 6 years of age with severe atopic dermatitis and for adolescents and adults with moderate-to-severe atopic dermatitis who are eligible for systemic therapy.
Dupilumab is a fully human monoclonal antibody that prevents the signaling of the interleukin-4 (IL-4) and interleukin-13 (IL-13) proteins, using Regeneron’s proprietary Veloclmmune technology. IL-4 and IL-13 are the central drivers of the type 2 inflammation, which plays a major role in atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyposis, and eosinophilic esophagitis. Dupilumab is currently approved in more than 60 countries and does not require ongoing lab monitoring, according to the press release.
This article was originally published by our sister publication
Reference:
1. Dupixent® (dupilumab) smpc updated with long-term data reinforcing well-established safety profile in adults with moderate-to-severe atopic dermatitis. Regeneron. Published June 28, 2021. Accessed June 30, 2021.