Dermatologic differences in a diverse population
Understanding the difference in biology and clinical presentations of cutaneous conditions in different skin types is critical for health care professionals to treat a diverse population.
Many different terms are used to describe skin color, from using actual colors to categorize individuals to identification based on one’s ethnicity. The most common terminology used in dermatology is the Fitzpatrick skin phototypes scale that classifies skin types from I (White) to VI (Black). This staging is graded by sensitivity or ability of the skin to tan with UV exposure.1 The term “skin of color” has been defined in many ways, but it typically includes individuals of Asian, Hispanic, Native American, Indian, and African descent.
Melanocytes in the basal layer of the epidermis produce cutaneous pigment known as melanin. Biological differences in different skin types underlie the overall color of the skin. For example, Black skin is naturally rich in melanin because melanosomes are distributed throughout the entire epidermis, whereas with fair White skin, few melanosomes are found at the stratum basale and melanosomes are absent in the upper layers of epidermis.
Understanding the difference in biology and clinical presentations of cutaneous conditions in different skin types is critical for health care professionals to treat a diverse population. The epidemiology, role of genetics, clinical presentation, and treatment considerations for atopic dermatitis, acne, and vitiligo in all skin types of the pediatric population are discussed here.
Atopic dermatitis
Atopic dermatitis (AD) is widely considered to be the most common chronic skin disease and typically starts in early infancy or childhood.2 Up to 25% of children and up to 8% of adults have a past or present diagnosis of AD.3 The pathology behind AD is thought to be multifactorial and likely includes a genetic predisposition combined with epidermal dysfunction, as well as environmental and socioeconomic factors.2 Additionally, AD is included in the “atopic diathesis,” along with food allergies, asthma, and allergic rhinitis (hay fever). Other comorbidities may include behavioral and psychosocial concerns.3
Epidemiology
Prevalence of AD in the United States is notably higher among Black children, who are 1.7 times more likely to be diagnosed with AD compared with White children.3 Furthermore, individuals of Asian descent are 7 times more likely to be diagnosed with AD than White individuals.4
Role of genetics
Filaggrin (FLG) loss-of-function mutations are currently the most-thoroughly studied genetic component of AD.4 Filaggrin is a major structural protein of the stratum corneum barrier, the outermost layer of the epidermis.4 Individuals with mutations of the FLG gene typically present with an earlier onset of atopic disease, more severe disease, and a higher risk of superinfection.4 Other polymorphisms in genes related to innate or adaptive immunity in Th2 signaling pathways are thought to play a role in the development of AD.4 Although psoriasis is thought to be related to issues in the Th1 signaling pathway including Th17 and interleukin 23 (IL-23), IL-17–positive T-cell expansion has been noted in Asian populations.5 Furthermore, it is postulated that the increased prevalence of AD seen in Asian populations may be due to a stronger Th17/22 polarization that may lead to a phenotype with AD-psoriasis overlap.4
Clinical presentation
Presentation of AD across all racial spectrums generally includes intense pruritus and xerosis accompanied by eczematous papules or plaques.6 However, subtleties of AD may present differently among various races. For example, skin lesions often appear more well demarcated in those of East Asian descent or may have greater extensor involvement in Black individuals.3 Additionally, xerosis and hyperlinear palms and soles may be more prominent in those with darker skin.3
Treatment considerations
Of note, the majority of studies concerning treatment of AD were conducted on White individuals, which may lead to difficulty in individualizing treatment regimens.4 Current guidelines support similar treatments for AD across all racial backgrounds, with first-line therapy including topical corticosteroids and calcineurin inhibitors.4 Additionally, one phosphodiesterase- 4 inhibitor, crisaborole, may be used for the treatment of mild to moderate AD.4 Anecdotally, topical steroids appear to be effective in all skin types; however, those of high potency should be approached with caution because they may lead to hypopigmentation particularly in skin of color.4 Systemic treatment can be considered for moderate to severe AD.
Acne
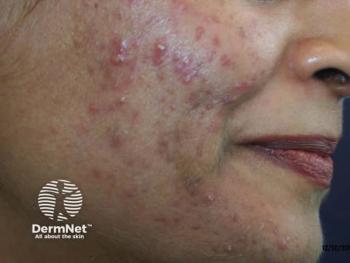
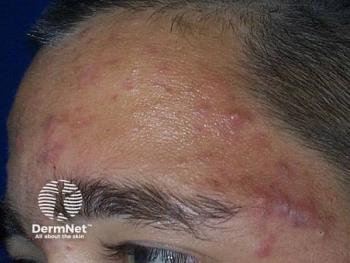
Acne is consistently a top 3 reported skin condition in the general population in the United States, United Kingdom, and France7 and is in the top 3 most common dermatoses in Black, Hispanic, and Asian adolescents.8,9 The pathophysiology of acne is multifactorial, beginning with a microcomedone and progressing to comedones. Follicular hyperkeratinization, increased sebum production, and proliferation of Cutibacterium acnes lead to subsequent follicular plugging, release of inflammatory cytokines, and activation of toll-like receptor 2.10 This process creates inflammatory papules, pustules, and nodules.10 The resulting sequelae involve various types of scarring as well as postinflammatory hyperpigmentation (PIH).
Epidemiology
Acne affects patients of many ages, but roughly 20% of young people suffer from moderate to severe acne.7 Some degree of acne affects almost all individuals aged 15 to 17 years, with an estimated 85% prevalence rate among individuals aged 12 to 24 years.7 A higher percentage of Hispanic teens have reported acne compared to Black and Asian teens.11 Frequency of acne in different ethnic groups has been studied, but results are mixed, largely due to difficulty with selection bias of study populations. Overall, prevalence and incidence appears to be similar among ethnic groups.7
Role of genetics
Little research is available regarding pathophysiologic differences among ethnic groups.7,8 Of the available research, most involve White and Black skin types, with less information available regarding acne in Asian and Hispanic skin types.7 There is no defined ethnic association or gene mutation in individuals with acne, although certain skin types are affected differently.
Heritability is almost 80% in firstdegree relatives. It tends to be more severe and occur earlier in patients with a positive family history of acne.7 Severity of acne also increases with maturity and dehydroepiandrosterone sulphate levels.7 It typically begins in early teen years with onset of facial sebum production.7 Of note, the follicular epithelium of the pilosebaceous unit is thicker in patients of skin type V and IV than skin types I and II. These follicles are more likely to keratinize and form comedones, rather than inflammatory and cystic lesions seen in patients with lighter skin types.9
Clinical presentation
Papular lesions are the most common type of acne lesion described in patients across all skin types.12 Postinflammatory hyperpigmentation and hyperpigmented macules most commonly affect darker skin types.9 Clinically, White and Asian patients tend to have more inflammatory acne than African American patients, who tend to have more comedonal acne.9 White, Hispanic, and Asian patients are more likely to present with nodulocystic acne compared with African American patients.9 In addition, Hispanic patients typically experience more pustular lesions and more scarring compared with Asian and African American patients, although African Americans experience more PIH.11 Asian and Middle Eastern patients report mostly papular and comedonal lesions and less PIH.12 Keloidal scarring is not a common complication of acne, but the risk is much greater in darker skin types.7,12
Treatment considerations
Several studies have shown significant differences in illness perception and quality of life in White vs non-White patients, specifically regarding White patients with acne experiencing less negative impact on social functioning compared to non-White patients with acne.13 A study by Barbieri et al showed that non-Hispanic Black patients were more likely to be seen by a dermatologist for acne compared with non-Hispanic White patients but were prescribed fewer acne medications.14 These papers, however, most commonly study adult patients, so much less literature is available regarding these concerns in the pediatric population.13 Regardless, this should be kept in mind when treating pediatric acne in all skin types.
In general, a thorough history of a patient’s skin type and sensitivity is necessary to formulate an appropriate treatment regimen. Hyperpigmentation, more common in patients of darker skin types, has been shown to be more distressing to many patients than the acne itself.12 Keep in mind that although comedones in Black patients do not appear red or inflamed clinically, there is a marked amount of histologic inflammation.9 This is especially seen in Black female patients.9 Postinflammatory hyperpigmentation can occur from the acne lesions themselves or from overly aggressive therapies that are irritating to the skin.12 Thus, care must be taken to avoid inducing irritation and retinoid dermatitis when starting acne therapy in patients with darker skin of color.8
Irritant contact dermatitis seems to be more prevalent in clinical practice in patients with darker skin of color, although the American Contact Dermatitis Society has studied this and found no difference in incidence between African American and White patients.11 Regardless, keep this in mind when selecting topical treatment regimens for this patient population.8,11 Vehicles such as lotions and creams should be utilized in patients with dry, sensitive skin prone to irritation, with methods such as slowly increasing frequency of product use at initiation. Also consider starting at low strength and slowly increasing the strength as tolerated.11 Other vehicles such as solutions and gels are more appropriate for patients with oily skin that is not prone to irritation.11
Vitiligo
Vitiligo is a disorder of depigmentation resulting from loss of melanocytes.15 Due to the potential disfiguring nature of the disease, psychosocial functioning can be impacted in both patients and their families.15 This is especially true in patients who develop vitiligo in childhood. The pathogenesis of vitiligo is not fully elucidated, but one of the most widely considered theories considers vitiligo to be autoimmune in nature, with the predominant cause of melanocyte loss due to attack by CD8-positive cytotoxic T cells.16
Epidemiology
The prevalence of vitiligo worldwide is approximately up to 2% of the population, including both children and adults.15 Up to 80% of patients develop vitiligo before aged 30 years, with up to 25% of patients developing their first depigmented lesion by the age of 10.17 The disease may impact all racial groups and skin types; however, there appears to be geographical differences regarding prevalence of disease.17 Women do appear to bemore commonly affected than men.17
Role of genetics
Genetic involvement in the development of vitiligo is thought to be very complex, and there is no specific genetic test for vitiligo.15 Involved genes are typically related to immune regulation, melanogenesis, and apoptosis.17 A familial association has been made with approximately 10% of patients having at least 1 first-degree relative also diagnosed with vitiligo.17
Clinical presentation
Lesions consistent with a diagnosis of vitiligo are amelanotic macules, possibly coalescing into larger depigmented patches, with distinct margins.17 Vitiligo is classified into nonsegmental, segmental, and undetermined.18 Nonsegmental vitiligo can then be further classified based on distribution.18 Additionally, halo nevi, melanocytic nevi surrounded by depigmented rims, may be visualized in individuals with vitiligo.19 Vitiligo is often associated with additional comorbid disorders.20 Comorbid autoimmune conditions are present in 10% to 15% of patients with vitiligo (thyroid disease is the most common.)20 Racial variation in autoimmune disease has been noted; hypothyroidism is most common in White patients; psoriasis is the most frequent in Asian patients. Systemic lupus erythematosus and rheumatoid arthritis are the most common comorbidities in Black patients.20
Treatment considerations
Due to the chronic and often progressive nature of vitiligo, treatment expectations must be clearly explained and discussed.19 Furthermore, treatment often spans several months and possibly years on an intermittent basis.19 All patients with a diagnosis of vitiligo should be encouraged to wear a daily sunscreen (SPF 30 or greater) due to the likelihood of burning depigmented skin.19 If desired, several brands of cosmetic camouflage and concealers exist and have demonstrated improvement in quality of life.19
Main treatment modalities for vitiligo include topical therapies and phototherapy. The 3 main classes of topical treatments include steroids, calcineurin inhibitors, and vitamin D.15 Phototherapy, mainly narrowband UV-B, is typically favored for widespread disease or disease that is rapidly progressive.1 Phototherapy may also be combined with use of topical steroids, which may increase the efficacy of UV-B.20
Conclusion
Dermatologic conditions have a wide variety of presentations across different skin types, with differing implications in prognosis and treatment. This variation is multifactorial, due to biological and environmental differences and cultural practices across individuals of different skin types. Clinical differences when treating a variety of skin of color involve pigmentary and inflammatory predilections and their sequelae. There are numerous etiological and treatment considerations for patients of differing skin types that providers should consider when evaluating this population. This is well described in darker skin types for certain dermatologic procedures such as cryotherapy and intralesional steroids.
The conditions discussed here are only a few of the many dermatologic conditions for which more research is needed in skin of color in the pediatric population. Little comprehensive research is available regarding many cutaneous diseases in the pediatric population—and even less regarding difference across skin types. More validated information is needed for better directed and individualized care for this vast patient population.
References:
1. Taylor SC. Skin of color: biology, structure, function, and implications for dermatologic disease. J Am Acad Dermatol. 2002;46(suppl 2):S41-S62. doi:10.1067/mjd.2002.120790
2. Brunner PM, Guttman-Yassky E. Racial differences in AD. Ann Allergy Asthma Immunol. 2019;122(5):449-455. doi:10.1016/j.anai.2018.11.015
3. Croce EA, Levy ML, Adamson AS, Matsui EC. Reframing racial and ethnic disparities in AD in Black and Latinx populations. J Allergy Clin Immunol. 2021;148(5):1104-1111. doi:10.1016/j.jaci.2021.09.015
4. Kaufman BP, Guttman-Yassky E, Alexis AF. AD in diverse racial and ethnic groups-variations in epidemiology, genetics, clinical presentation and treatment. Exp Dermatol. 2018;27(4):340-357. doi:10.1111/exd.13514
5. Noda S, Suárez-Fariñas M, Ungar B, et al. The Asian atopic dermatitis phenotype combines features of AD and psoriasis with increased TH17 polarization. J Allergy Clin Immunol. 2015;136(5):1254-1264. doi:10.1016/j.jaci.2015.08.015
6. Monir RL, Schoch JJ, Garvan CW, Neu J, Lemas DJ. Association between atopic dermatitis and race from infancy to early childhood: a retrospective cohort study. Int J Dermatol. Published online August 11, 2021. doi:10.1111/ijd.15805
7. Bhate K, Williams HC. Epidemiology of acne vulgaris. Br J Dermatol. 2013;168(3):474-485. doi:10.1111/bjd.12149
8. Maymone MBC, Watchmaker JD, Dubiel M, Wirya SA, Shen LY, Vashi NA. Common skin disorders in pediatric skin of color. J Pediatr Health Care. 2019;33(6):727-737. doi:10.1016/j.pedhc.2019.04.019
9. Halder RM, Brooks HL, Callender VD. Acne in ethnic skin. Dermatol Clin. 2003;21(4):609-615, vii. doi:10.1016/s0733-8635(03)00082-2
10. Ashton R, Weinstein M. Acne vulgaris in the pediatric patient. Pediatr Rev. 2019;40(11):577-589. doi:10.1542/pir.2018-0137
11. Taylor SC, Cook-Bolden F, Rahman Z, Strachan D. Acne vulgaris in skin of color. J Am Acad Dermatol. 2002;46(suppl 2):S98-S106. doi:10.1067/mjd.2002.120791
12. Morrone A, Franco G, Valenzano M, et al. Clinical features of acne vulgaris in 444 patients with ethnic skin. J Dermatol. 2011;38(4):405-408. doi:10.1111/j.1346-8138.2010.00976.x
13. Nagpal N, Gordon-Elliott J, Lipner S. Comparison of quality of life and illness perception among patients with acne, eczema, and psoriasis. Dermatol Online J. 2019;25(5):13030/qt3fk3f989.
14. Barbieri JS, Shin DB, Wang S, Margolis DJ, Takeshita J. Association of race/ethnicity and sex with differences in health care use and treatment for acne. JAMA Dermatol. 2020;156(3):312-319. doi:10.1001/jamadermatol.2019.4818
15. Ezzedine K, Silverberg N. A practical approach to the diagnosis and treatment of vitiligo in children. Pediatrics. 2016;138(1):e20154126. doi:10.1542/peds.2015-4126
16. Dina Y, McKesey J, Pandya AG. Disorders of hypopigmentation. J Drugs Dermatol. 2019;18(3):s115-s116.
17. Bergqvist C, Ezzedine K. Vitiligo: a review. Dermatology. 2020;236(6):571-592. doi:10.1159/000506103
18. Nicolaidou E, Mastraftsi S, Tzanetakou V, Rigopoulos D. Childhood vitiligo. Am J Clin Dermatol. 2019;20(4):515-526. doi:10.1007/s40257-019-00430-0Nicolaidou E, Mastraftsi S, Tzanetakou V, Rigopoulos D. Childhood vitiligo. Am J Clin Dermatol. 2019;20(4):515-526. doi:10.1007/s40257-019-00430-0[GC1]
19. Dahir AM, Thomsen SF. Comorbities in vitiligo: comprehensive review. Int J Dermatol. 2018;57(10):1157-1164. doi:10.1111/ijd.14055
20. Boniface K, Seneschal J, Picardo M, Taïeb A. Vitiligo: focus on clinical aspects, immunopathogenesis, and therapy. Clin Rev Allergy Immunol. 2018;54(1):52-67. doi:10.1007/s12016-017-8622-7