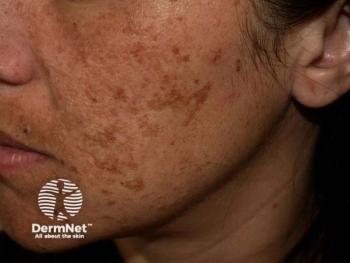
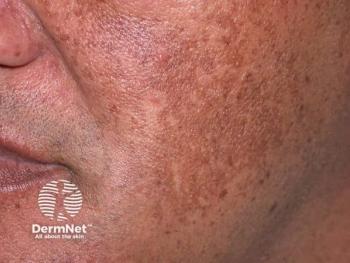
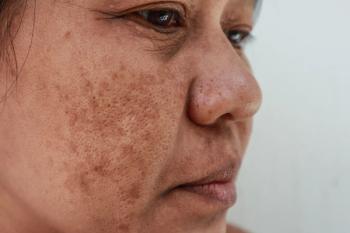
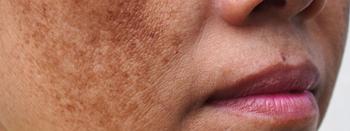
Combination treatment outshines oral treatment alone for melasma
Oral tranexamic acid combined with low fluence 1064 nm Q-switched Nd: YAG laser is an effective and safe melasma treatment, with outcomes that are better than oral tranexamic acid alone, according to a recent study.
Oral tranexamic acid combined with low fluence 1064 nm Q-switched Nd: YAG laser is an effective and safe melasma treatment, with outcomes that are better than oral tranexamic acid alone, according to
Studies suggest that tranexamic acid, a trans-4-(aminomethyl) cyclohexane carboxylic acid, modulates melasma’s vascular component and inhibits melanin synthesis. The high-intensity beam and short pulses from the Q-switched Nd: YAG 1064 nm laser target melanin in melanocytes, keratinocytes or dermal melanophages, removing brown dyschromia and red vascularity, according to the authors.
“Though there are many lines of melasma treatment, in the absence of any ‘gold standard,’ it still remains a challenge for dermatologists,” the authors write.
To compare tranexamic acid oral monotherapy with oral tranexamic acid and laser treatment for melasma, researchers in Greece and Egypt studied 60 female melasma patients. Thirty women (group A) received 250 mg daily of oral tranexamic acid for three months, while 30 women (group B) received the oral treatment along with Q-switched Nd: YAG 1064 nm laser treatments every two weeks for three months, unless they improved before three months. Researchers kept the energy level for laser treatment to a minimum of 2 J/cm2 to avoid side effects.
The researchers found that Modified Melasma Area and Severity Index (mMASI) improved significantly compared to baseline in both groups, but group B’s response was notably higher than that of group A. Eighteen, or 60%, of the patients in group B achieved melasma clearance before completing three months of laser treatments.
“Epidermal type of melasma showed the best response (0.048) and telangiectasias significantly improved in both groups,” they write.
They report the percent change in mMASI score post treatment was highest in melasma patients with skin type III and lowest in skin type V.
At baseline, 20 patients in group A and 24 in group B had telangiectasia. Post treatment, only nine patients in group A and four in group B had telangiectasia.
Side effects in both groups were minimal, transient and tolerable, according to the authors. Group B was more likely to experience complications due to the laser. These included patients with long-standing erythema, itching and punctate leukoderma.
Researchers followed patients for three months post treatment for recurrence and found that melasma recurred in three patients in group A and one in group B.
Studies looking at the use of lasers to treat melasma have had mixed results. Some found adding the laser to oral tranexamic acid resulted in post-inflammatory hyperpigmentation, while others found that results from the Q-switched Nd: YAG in Asian melasma patients were temporary. However, researchers reported in
Further research is needed on long-term administration of tranexamic acid, as well as how to further reduce melasma recurrence rate, according to the authors.
Disclosures:
The authors report no conflicts of interest.
References:
Agamia N, Apalla Z, Salem W, Abdallah W. A comparative study between oral tranexamic acid versus oral tranexamic acid and Q-switched Nd-YAG laser in melasma treatment: a clinical and dermoscopic evaluation. J Dermatolog Treat. 2020 Jan 7:1-8. doi: 10.1080/09546634.2019.1708847.
Newsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.