- General Dermatology
- Eczema
- Alopecia
- Aesthetics
- Vitiligo
- COVID-19
- Actinic Keratosis
- Precision Medicine and Biologics
- Rare Disease
- Wound Care
- Rosacea
- Psoriasis
- Psoriatic Arthritis
- Atopic Dermatitis
- Melasma
- NP and PA
- Skin Cancer
- Hidradenitis Suppurativa
- Drug Watch
- Pigmentary Disorders
- Acne
- Pediatric Dermatology
- Practice Management
94% of Patients With Atopic Dermatitis Report Satisfaction With Baricitinib Treatment
Approximately 76% of patients reported being "satisfied," while 18% reported they were "very satisfied" with treatment.
Ольга Тернавская/Adobe Stock
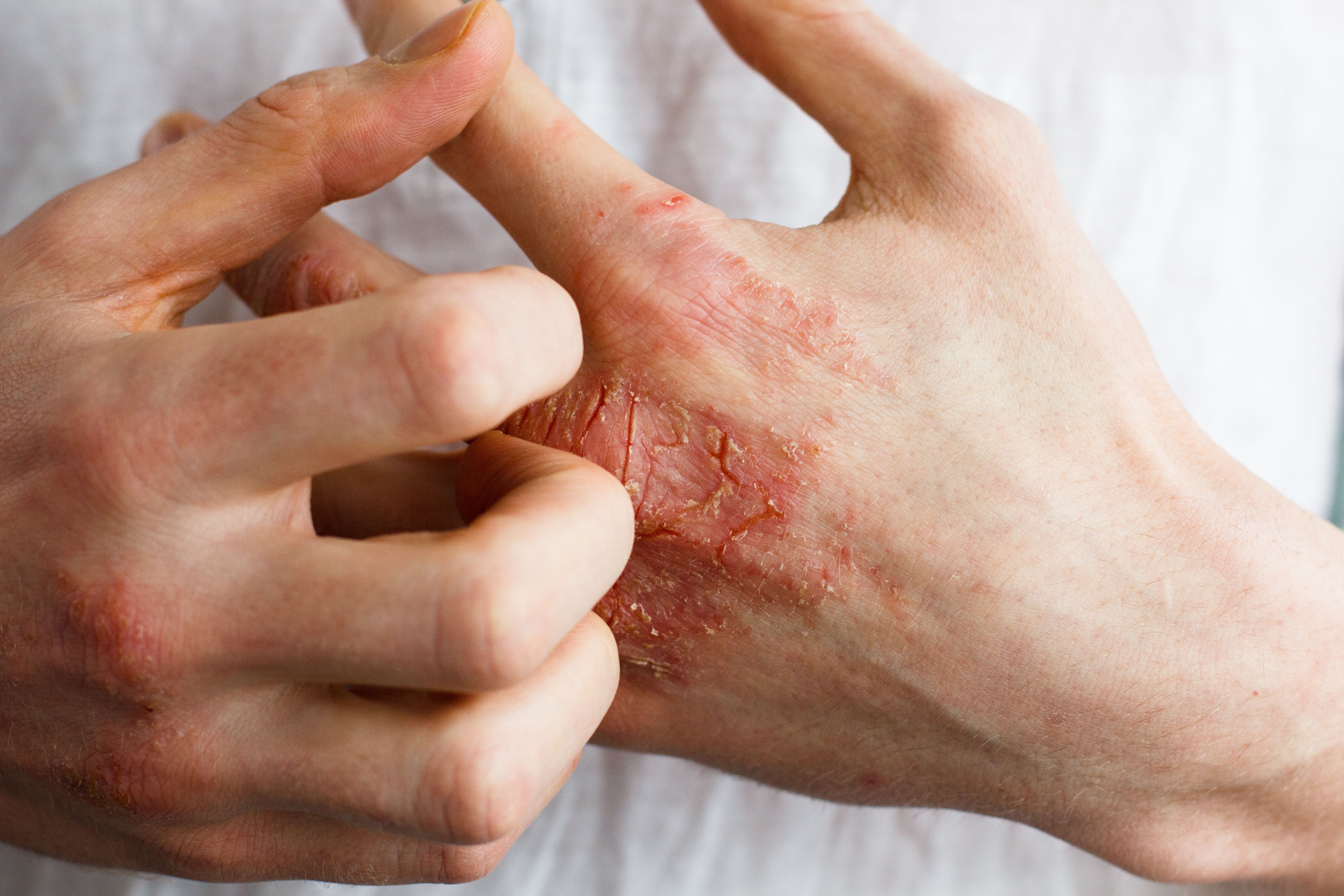
Most patients with atopic dermatitis (AD) reported being either satisfied or very satisfied with baricitinib treatment, according to a study published in the Journal of Dermatological Treatment.
Despite its benefits, the researchers explained that there is currently no data on treatment satisfaction in patients with moderate to severe AD following initiation of any oral JAK inhibitor. Consequently, the researchers conducted a study to explore patients' experiences with baricitinib in real-world clinical settings. They assessed factors contributing to treatment satisfaction and patient-reported AD outcomes among those treated with baricitinib.
Data were collected between June 6, 2022, and January 16, 2023, through a one-time online structured questionnaire with close-ended questions designed to meet the study objectives. Eligible patients were 18 years or older with moderate to severe AD treated with baricitinib in clinical practice for 4 or more weeks in Germany, France, or the United Kingdom; participants were recruited through their managing health care practitioner (HCP).
170 patients treated with baricitinib completed the survey, 69 from the United Kingdom, 53 from Germany, and 48 from France. At treatment initiation, the mean (SD) patient age was 39.3 (13.5) years; 59% were female, and the mean (SD) time since AD diagnosis was 20.9 (14.0) years.
The survey consisted of a series of questions related to patient satisfaction with baricitinib that the researchers measured on a 4-point Likert scale, with potential responses being “very satisfied,” “satisfied,” “dissatisfied,” and “very dissatisfied.” Also, the medication-generic Abbreviated Treatment Satisfaction Questionnaire for Medication-9 (TSQM-9) was used to measure global patient satisfaction with baricitinib; the TSQM-9 ranges from 0 to 100, with higher scores indicating higher satisfaction.
At baricitinib initiation, most patients rated their AD as either “severe” (79%) or “moderate” (19%). Patients also experienced high itch levels at initiation with a mean (SD) Itch Numerical Rating Scale (NRS) of 6.8 (2.0).
Based on the Likert scale, most patients using baricitinib reported being either “satisfied” (76%) or “very satisfied” (18%). In particular, many patients reported being “satisfied” (56%) or “very satisfied” (36%) with its itch control; patients also reported being either “very satisfied” (21%) or “satisfied” (69%) with how quickly they saw results.
Using the TSQM-9 to evaluate baricitinib, patients reported a mean (SD) global satisfaction score of 62.7 (20.5) and a mean (SD) effectiveness score of 68.1 (15.3). Additionally, they reported a mean (SD) TSQM-9 convenience score of 78.0 (14.0) as patients described baricitinib as “very/extremely convenient” (57%) and “very/extremely easy” (64%) to use.
Because baricitinib helped to control skin inflammation, many patients reduced the co-application of topical AD treatments. For those who used TCS, 50% reported reducing their use while 32% reported discontinuing it entirely after using baricitinib. As for those who used TCI, 22% reported reduced usage while 31% reported discontinuing use completely. Also, 44% of patients using emollients reported using less since they began using baricitinib.
The researchers acknowledged their study’s limitations, one being that the sample size may limit the generalizability of their findings. Also, they explained that some data was gathered from patients’ retrospective observations, so recall bias is an issue that may have affected their findings. Lastly, only so much could be explored as the survey was conducted during one interaction rather than several time points; because of this, the researchers urged others to build upon their findings.
“Future research in this area could usefully examine several questions that were outside the scope of our research, including examining the relationship, if any, between patient-reported outcomes and treatment satisfaction, allowing for sub-analyses of treatment satisfaction by topographic site, and examining what drove discontinuation of prior lines of therapy before the initiation of baricitinib,” the authors wrote.
Reference
Augustin M, Nosbaum A, Werfel T, et al. Treatment satisfaction of adult patients with moderate-to-severe atopic dermatitis treated with baricitinib in France, Germany, and the United Kingdom: results from a cross-sectional international patient survey. J Dermatolog Treat. 2023;34(1):2276047. doi:10.1080/09546634.2023.2276047
[This article was originally published by our sister publication, American Journal of Managed Care.]
