Ustekinumab Approved by the FDA for Pediatric Patients with Psoriatic Arthritis
Janssen Pharmaceutical Companies of Johnson & Johnson announced the approval of ustekinumab as a new treatment option for patients aged 6 years or older with active psoriatic arthritis.
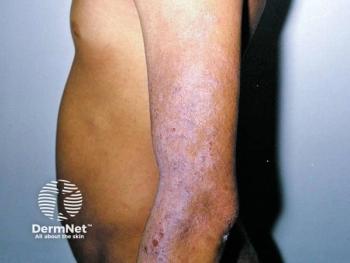
The US Food and Drug Administration (FDA) has approved ustekinumab (Stelara; Janssen Pharmaceutical) for the treatment of active psoriatic arthritis (PsA) in pediatric patients. Ustekinumab is the first and only biologic that targets both cytokines interleukin (IL)-12 and IL-23.1 PsA is a rare disease that mimics adult PsA and affects 5%-8% of children and adolescents that suffer from chronic inflammatory arthritis.
Since the first approval for adults with moderate to severe PsA in 2009, 2 out of the 4 indications for ustekinumab now include pediatric patients to further expand its treatment reach. Ustekinumab is a fully monoclonal antibody that selectively inhibits interleukin (IL)-12 and IL-23. Medical professionals recognize that both cytokines are important to calm down the overactive inflammatory response in many autoimmune diseases.
Ustekinumab is injected subcutaneously 4 times per year after 2 initial starter doses. The approval is based on pharmacokinetic (PK) data and the conclusion of the efficacy and safety of ustekinumab in multiple phase 3 studies in adult and pediatric patients with moderate to severe plaque psoriasis (PsO) (PSTELLAR, CADMUS, and CADMUS Jr) and adult patients with active PsA (PSUMMIT I and II). Data showed that PK exposure of ustekinumab in pediatric PsO patients with active PsA was consistent with phase 3 clinical trials of ustekinumab in pediatric PsO patients without active PsA, as well as with adult patients with moderate to severe PsO or adult patients with active PsA. Data on similar efficacy endpoints were similar in the pediatric PsO patients with active PsA.
Symptoms of pediatric PsA include joint inflammation and skin lesions. Overall, pediatric PsA is difficult to treat and needs a standardized treatment option. Some common adverse events of ustekinumab include nasal congestion, sore throat, runny nose, headache, itching, stomach pain, and joint pain.
Reference
- Johnson JPC of J&J. Stelara (Ustekinumab) approved by the US food and drug administration to treat pediatric patients with active psoriatic arthritis. Accessed August 1, 2022.
https://www.prnewswire.com/news-releases/stelara-ustekinumab-approved-by-the-us-food-and-drug-administration-to-treat-pediatric-patients-with-active-psoriatic-arthritis-301596940.html
Newsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.