Study: Daylight PDT vs Conventional PDT for Actinic Keratosis Treatment
Researchers examine whether daylight PDT is less painful but equally as effective as conventional PDT as treatment for actinic keratosis.
A research article in the
The investigators used Embase and PubMed to search for clinical articles that were published on the efficacy of daylight-PDT using the keywords “daylight photodynamic therapy” and “actinic keratosis.” There were no limits placed on the search time frame, however, it only included studies published before May 22, 2019. The studies were required to be in English and involve humans. A total of 9 relevant studies were included in the review.
cPDT uses either 5-aminolaevulinic acid (ALA) or methyl ester (MAL) as a topical photosensitizer to the affected area with AK. ALA is an early intermediate in the heme biosynthesis pathway and is also a precursor to the natural photosensitizer protoporphyrin IX (PpIX), according to the article. ALA non-selectively enters the cells, but the PpIX tends to accumulate in tumor cells. A blue or red light is then used to incubate the application.
“The standard procedure for cPDT calls for occlusive treatment with a photosensitizer for 1 to 3 hours, allowing for enough time for accumulation of PpIX,” the authors wrote. “However, FDA approval for use of photosensitizers in the treatment of AK involves incubation periods of 3 hours for MAL, 18 hours for ALA, and 3 hours for nanoemulsions for ALA.”
After cPDT, patients report pain as the most common adverse event (AE). The discomfort of treatment, frequency of treatment, and the length of the procedures lead to the search of more tolerable PDT.
For daylight-PDT to be considered a reasonable alternative to cPDT, it has to be effective in the short and long term, according to the study.
A meta-analysis of 6 randomized controlled trials that compared daylight-PDT and cPDT found daylight-PDT is noninferior to cPDT in the treatment of grade I and II AKs with 2 of the studies included in the analysis randomized, controlled, investigator-blinded, split-body, and multicenter. They were based in Australia and Europe and included 100 patients with grade I and II AKs.
Both studies excluded individuals with severe AK with 2 hours of daylight exposure time 30 minutes after MAL application. The Australian study demonstrated a complete lesion response rate of 89.2% and 92.8% for daylight-PDT and cPDT, respectively, while the European study resulted in complete lesion response rates of 70% for daylight-PDT and 74% for cPDT. Daylight-PDT demonstrated noninferiority to cPDT at 12-week follow-up in both studies. After 6 months, the Australian study found that both daylight-PDT and cPDT maintained lesion clearance for 96% of grade I lesions.
For more details on the length of exposure and the joules of exposure
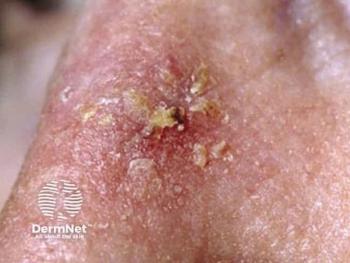
While efficacious, the authors wrote, daylight-PDT is less effective in moderate to thick AKs compared to thick AKs. A study found that at 3 months following daylight-PDT treatment, 73% of grade I lesions showed a complete response, compared to 63% of grade II lesions and 55% of grade III lesions. Twenty three percent of grade II and 39% of grade III lesions were reduced to a lower lesion grade. Also, weather (cloudy or sunny days) does not seem to be an issue for treatment, however going out in rainy weather should be avoided, researchers noted.
According to the article, 2 studies show that ALA may be slightly more effective than MAL.
A randomized observer-blinded study with a split-face trial of 13 patients and a collective total of 177 grade I–III AKs compared daylight-PDT treatments using ALA nanoemulsion gel or MAL cream as the photosensitizer. The trial had grade I AKs treated once and grade II–III AKs treated twice with a 0.25-mm layer of photosensitizer. Overall, the nanoemulsion ALA daylight-PDT cleared 85% of the AKs vs MAL daylight-PDT cleared 74% of the total AKs (P =.099).
Another study compared BF‐200 ALA—a nanoemulsion gel containing 7.8% ALA—to MAL in the treatment of mild to moderate AK with daylight-PDT in 52 patients from 7 centers in Germany and Spain. Similar clearance rates were seen in the 12 weeks following treatment for BF-200 ALA gel and MAL (79.8 ± 23.6% and 76.5 ± 26.5%, respectively, P < .0001). The sides treated with BF‐200 ALA gel also had lower 1‐year lesion recurrence rates (19.9%) compared with sides treated with MAL cream (31.6%) (P = .01).
“Pretreatment of the skin prior to treatment with daylight-PDT is suggested to improve efficacy by increasing penetration of the ALA or MAL,” the authors wrote.
Treatment guidelines from groups of experts in both Europe and Australia have been independently published, but none for the United States.
Common AEs of daylight-PDT are mild and include erythema, blistering, itching, and crusting, with scarring being a rare occurrence.
Current evidence suggests that daylight-PDT is a safe and effective alternative to cPDT for thin to moderate AK lesions, according to the authors, but further studies are needed to determine the minimum exposure time necessary to minimize side effects. Also, physicans should be aware that daylight-PDT is a potential treatment option for patients with low pain tolerance or limited scheduling.
Disclosure:
Raja K Sivamani serves as a medical editor for LearnHealth and as a consultant for Burt’s Bees, Dermala, and Tomorrow’s Leaf. The authors report no other conflicts of interest.
Reference:
Nguyen M, Sandhu SS, Sivamani RK. Clinical utility of daylight photodynamic therapy in the treatment of actinic keratosis – a review of the literature. Clin Cosmet Investig Dermatol. 2019;12:427-435. doi:10.2147/CCID.S167498
Newsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.