- General Dermatology
- Eczema
- Alopecia
- Aesthetics
- Vitiligo
- COVID-19
- Actinic Keratosis
- Precision Medicine and Biologics
- Rare Disease
- Wound Care
- Rosacea
- Psoriasis
- Psoriatic Arthritis
- Atopic Dermatitis
- Melasma
- NP and PA
- Skin Cancer
- Hidradenitis Suppurativa
- Drug Watch
- Pigmentary Disorders
- Acne
- Pediatric Dermatology
- Practice Management
Pelthos Therapeutics Launches to Accelerate Commercialization of Treatment for Molluscum Contagiosum
The new launch is set to bring berdazimer topical gel, 10.3% to adults and pediatric patients over the age of 1 with molluscum contagiosum faster.
Molluscum contagiosum skin lesions on a child (ZayNyi/Adobe Stock)
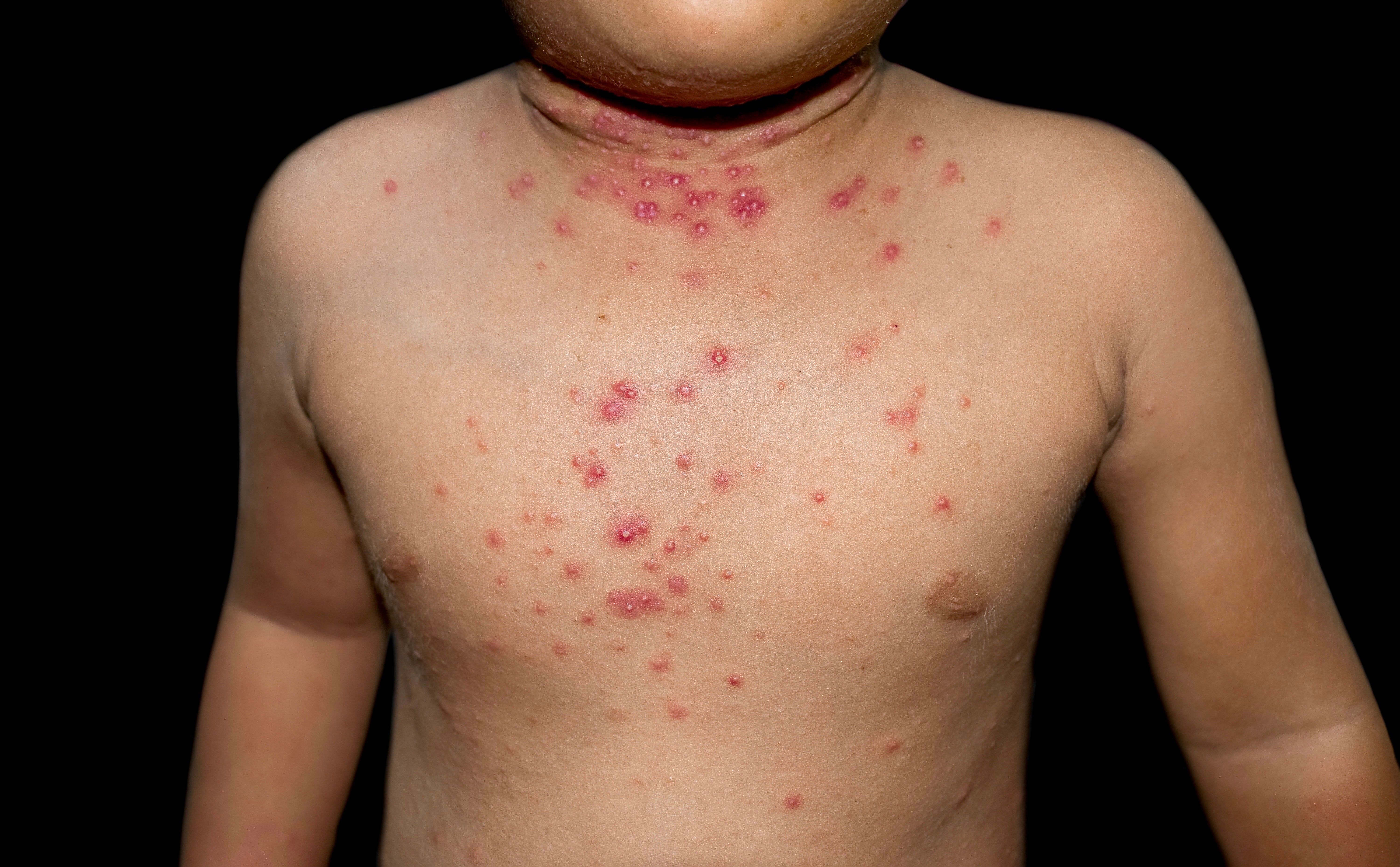
Ligand Pharmaceuticals (Nasdaq: LGND) has unveiled Pelthos Therapeutics (Pelthos), a biopharmaceutical endeavor dedicated to bringing forth therapeutic solutions to patients in need. One of the first initiatives of the Pelthos launch is to accelerate the commercialization of berdazimer topical gel, 10.3% (Zelsuvmi) approved to treat molluscum contagiosum in adults and pediatric patients over the age of 1. Scott Plesha, CO of Pelthos Therapeutics, believes the launch of the company will mark a significant stride towards addressing unmet medical needs in dermatology.1
“I am excited to assume the leadership of Pelthos as we prepare to offer the first at-home therapy for the many patients who are completely untreated today or who need a more suitable option,” said Plesha in a news release. “This is a company that has several meaningful milestones on the horizon and a team that is ready to execute to meet the needs of patients.”
Pelthos pledges to introduce safe and efficacious therapeutic products aimed at assisting patients grappling with diseases that currently lack adequate treatment options. The focal point of the company's efforts isberdazimer topical gel, 10.3%, as it holds a Novel Drug designation from the US Food and Drug Administration (FDA), positioning it as the inaugural and sole approved topical prescription medication for at-home application.The NDA and approval are based on positive data stemming from the B-SIMPLE4 (NCT04535531) trial, published in the Journal of the American Medical Association Dermatology. In the phase 3 study, berdazimer gel demonstrated statistically significant improvements in the clearance of treatable molluscum contagiosum at week 12. Key outcomes of the study also included a lesion count of 0 or 1 of all treatable molluscum contagiosum at week 12, a 90% reduction from baseline in the number of all treatable molluscum contagiosum at week 12, complete clearance of all treatable molluscum contagiosum at week 8, and a change from baseline in the number of all treatable molluscum contagiosum at week 4.2
This drug is underpinned by Pelthos' proprietary nitric oxide-based technology platform, Nitricil. Berdazimer topical gel, 10.3%, is poised for availability in the US by late 2024, promising a paradigm shift in molluscum contagiosum management.Pelthos' commitment extends beyond berdazimer topical gel, 10.3% with a pipeline of potential drug candidates leveraging the Nitricil platform to tackle diverse dermatological challenges.
The January 2024 approval of berdazimer gel is the second FDA-approval for molluscum contagiosum. “The approval of [berdazimer gel] Zelsuvmi is a breakthrough, marking the first time that clinicians can treat molluscum with an efficacious topical prescription medication that is applied by the patient, or a family member,” said Mark D. Kaufmann, MD, FAAD, clinical professor of Dermatology in the department of dermatology at the Icahn School of Medicine at Mount Sinai in New York, New York and past president of the American Academy of Dermatology, in a press release earlier this year. “I look forward to having this novel medication to treat my molluscum patients.”3
Currently, patients with molluscum contagiosum require a physician visit. Cantharidin topical solution 0.7% (Ycanth; Verrica PHarmaceuticals) was approved by the FDA for the treatment of molluscum contagiosum in adults and pediatric pages aged 2 years and older on July 21, 2023. The approval was based on positive data from 2 identical phase 3 randomized, double-blind, multicenter clinical trials (CAMP-1 and CAMP-2). The exclusive drug-device combination solution features a carefully regulated formulation of cantharidin (0.7% w/v) administered through a disposable applicator.2 Ycanth was launched commercially in September 2023, and the official J-code (J7354) launched by the US Centers for Medicare & Medicaid Services earlier this week.4
Have you utilized the latest treatment options for molluscum contagiosum? Would your patients benefit from an at-home option? What do you think of the current treatment landscape available? We would love to hear from you. Email us at DTEditor@mmhgroup.com.
References
- Ligand Pharmaceuticals announces launch of Pelthos Therapeutics to accelerate commercialization of ZELSUVMI™. News Release. Ligand Pharmaceuticals. April 3, 2024. Accessed April 3, 2024.https://investor.ligand.com/news-and-events/press-releases/news-details/2024/Ligand-Pharmaceuticals-Announces-Launch-of-Pelthos-Therapeutics-to-Accelerate-Commercialization-of-ZELSUVMI/default.aspx
- Browning JC, Enloe C, Cartwright M, et al. Efficacy and safety of topical nitric oxide-teleasingberdazimer gel in patients with molluscum contagiosum: a phase 3 randomized clinical trial. JAMA Dermatol. 2022 Aug 1;158(8):871-878. doi: 10.1001/jamadermatol.2022.2721. PMID: 35830173; PMCID: PMC9280611.
- Verrica Pharmaceuticals Announces FDA Approval of YCANTH (cantharidin) topical solution 0.7%. Verrica Pharmaceuticals. July 21, 2023. Accessed January 29, 2024. https://verrica.com/press_release/verrica-pharmaceuticals-announces-fda-approval-of-ycanth-cantharidin-topical-solution-0-7/
- Verrica Pharmaceuticals Receives Permanent J-Code (J7354) for YCANTH from Centers for Medicare and Medicaid Services. GlobeNewswire. January 29, 2024. Accessed January 29, 2024. https://www.globenewswire.com/news-release/2024/01/29/2818804/0/en/Verrica-Pharmaceuticals-Receives-Permanent-J-Code-J7354-for-YCANTH-from-Centers-for-Medicare-and-Medicaid-Services.html
