Dermatologist says topical therapies safe for atopic dermatitis tx
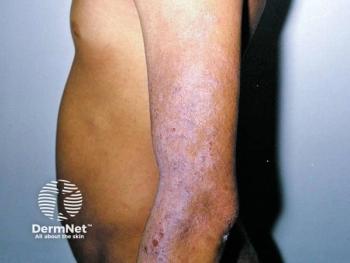
Portland, Ore. - Recently concerns have arisen regarding chronic use of the calcineurin inhibitors, Elidel cream (Novartis) and Protopic ointment (Astellas), in treating atopic dermatitis because of their potential association with infection and malignancy.
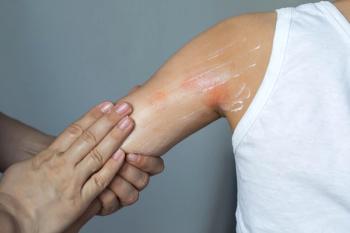
"These drugs should be used in a practical way," says Jon Hanifin, M.D., professor of dermatology, Oregon Health and Science University, University of Oregon. "They have given us a whole new dimension for treating bad atopic dermatitis, where we can maintain control for long periods of time because this is a chronic disease."
"Topical steroids, which are more potent, are the first-line drug. However, they can't be used every day," he says. "They are the way to shut off the inflammation quickly, and then for the long-term we can't use those steroids more than twice a week. So in between, we often need to use agents like Elidel and Protopic."
For this reason, the calcineurin inhibitors are the most effective secondary therapy. The Food and Drug Administration (FDA) recommends that healthcare providers, patients and caregivers consider using Elidel and Protopic only as second-line agents for short-term and intermittent treatment of atopic dermatitis in patients unresponsive to, or intolerant of, other treatments.
The agency also recommends that physicians avoid use of Elidel and Protopic in children younger than 2 years of age because the effect of Elidel and Protopic on the developing immune system in infants and children is not known.
The minimum amount of Elidel or Protopic needed to control the patient's symptoms should be used. In animals, increasing the dose resulted in higher rates of cancer, according to the FDA.
Clinical use
"I generally start the patient on a mid-strength topical steroid, and after anywhere from three to 10 days after the inflammation is controlled, I tell them to start using the calcineurin inhibitors twice a day," Dr. Hanifin says.
"Then, if they can taper it down to once a day, or every other day that is great," he says. "If the patient needs to add a steroid once or twice a week, that is quite safe."
In general, patients do not use the calcineurin inhibitors constantly. For one thing, the drugs are very expensive, so very few patients use them excessively.
According to Dr. Hanifin, with moderate use, safety is not an issue.
"There are theoretical risks of infection and long-term cancer, both skin cancer and lymphoma, if it is absorbed," he says. "For example, in those rare instances where people were using it all over their body, day after day, that may predispose to cancer."
Dr. Hanifin points out that he has seen no infection risk. "There was one study in a long-term trial that showed a very slight statistical increase in herpes simplex infection in people treated with calcineurin inhibitors. However, it really is not a relevant problem," he adds.
The FDA issued the warning to address the way drug companies were promoting the agents, especially to pediatricians, as first-line drugs, Dr. Hanifin explains.
Newsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.