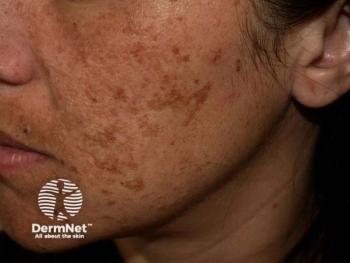
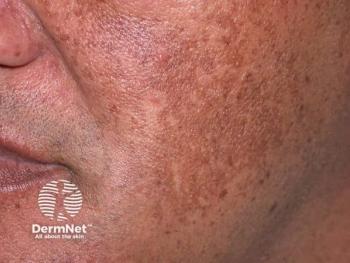
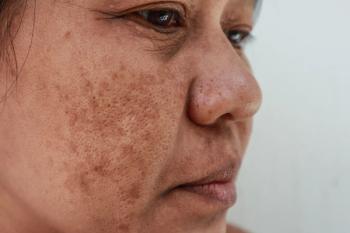
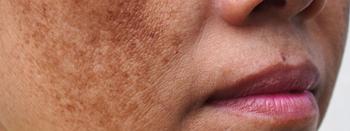
Cysteamine cream, tranexamic acid mesotherapy reveal similar efficacy, different safety
Cysteamine cream and tranexamic acid mesotherapy demonstrated similar efficacy but yielded different safety results when being compared as a treatment for melasma, according to a recently published study.
Researchers comparing melasma treatment cysteamine 5% cream to tranexamic acid mesotherapy found both were similarly effective but cysteamine cream appears to be safer.
The single-blind, randomized study of 54 Iranian patients with melasma was published Sept. 2 in the
Melasma treatment involves reducing melanocyte proliferation and inhibiting melanosome formation.“[Tranexamic acid] inhibits plasmin activity by preventing the adhesion of plasminogen to keratinocyte resulting in reduced prostaglandin and ultimately decreased melanocyte tyrosine kinase activity,” the authors wrote.
The simplest aminothiol in cells, cysteamine, has antioxidant properties and has long been shown in studies as a potent depigmenting molecule, according to the paper.
Researchers compared the treatments in a study of predominately female patients presenting to an Iranian dermatology clinic between November 2018 and July 2019. Those included in the study had suffered from melasma for at least a year and had not treated the condition with medication for at least two months.1
Over the course of four months, participants randomly received cysteamine 5% cream, which they were told to apply to melasma lesions after washing and drying their faces 30 minutes before bedtime.
Dermatologists performed mesotherapy on patients randomized to the other group using intradermal microinjections tranexamic acid mesotherapy, 0.05 mL (4 mg/mL). They delivered the injections using a 28-gauge needle at a depth of 1 mm with 10 mm distance to three areas. Dermatologists treated patients in the tranexamic acid group every four weeks, up to two months.1
To assess healing, the authors used the Dermacatch device (Colorix), which uses reflective light to measure melanin and erythema, and the modified Melasma Area Severity Index (mMASI).
Most of the patients studied, 29, had moderate melasma at baseline, with 12 being considered mild and 13 severe.1
They found melasma lesions improved by a rate of 32.5% at the second visit and 48% at the third visit in the tranexamic acid group. These rates were not significantly different in the cysteamine group, according to the study.1
“The most remarkable improvement rate was observed in the [tranexamic acid] group at the third visit based on mMASI and Dermacatch values at 47% and 15% in turn,” they wrote.
Dermacatch values fell, signaling improvement, at the second and third visits similarly in both groups.
Complications, however, differed between groups with cysteamine appearing safer than tranexamic acid mesotherapy. Researchers found tranexamic acid patients were significantly more likely to have treatment-related erythema, itching, burning and irritation compared to those treated with cysteamine. For example, among patients in the cysteamine group, 20 experienced no erythema, five had mild erythema, two had moderate and none of the patients in the group had severe erythema. Conversely, none of the patients in the tranexamic group were free of erythema. Twenty had mild, four moderate and three severe erythema as a result of treatment.
Most patients, 20, treated with cysteamine reported no itching, compared to 21 patients who reported mild itching in the tranexamic acid group. There was no notable difference in dryness between the groups.
“Overall, patients in the cysteamine group were in the none or mild categories of complications as compared to the [tranexamic acid] mesotherapy group,” they wrote. “It is worth noting that no recurrences of melasma were witnessed during the four months and eight weeks’ follow-up period for cysteamine and [the tranexamic acid] group, respectively.”
It is good to have options that show efficacy in melasma treatment because often used tyrosinase inhibitors, such as hydroquinone, azelaic acid and arbutin, result in common side effects from photosensitivity to pruritus and scaling, according to the authors.
Larger, carefully designed studies looking at determining optimal dosage, application frequency, benefits and sustained effects from these treatments should follow. Researchers should also conduct similar studies with longer follow ups to determine rate of relapse or tapering, they wrote.
Melasma prevalence in Arab-American populations is 13.4 to 15.5%, according to a study published July-August in the
These researchers compared the efficacy of tranexamic acid mesotherapy to 0.9% normal saline for melasma in a split-face study. The 12-week study suggests hemi-modified Melasma Area and Severity Scoring (H-mMASI) was significantly reduced from an average 3.19 to 1.52 on the tranexamic side compared to a decline in scores on placebo side.2
“Tranexamic Acid … mesotherapy can be considered as the most cost-effective, safe and directly observed therapy for melasma which showed significant improvement when old prior therapies have failed,” they concluded.
Reference:
1. Karrabi, M., Mansournia, M.A., Sharestanaki, E. et al. Clinical evaluation of efficacy and tolerability of cysteamine 5% cream in comparison with tranexamic acid mesotherapy in subjects with melasma: a single-blind, randomized clinical trial study. Arch Dermatol Res (2020).
2. Kaleem S, Ghafoor R, Khan S. Comparison of efficacy of Tranexamic Acid Mesotherapy versus 0.9% normal Saline for Melasma; A split face study in a Tertiary Care Hospital of Karachi. Pak J Med Sci. 2020;36(5):930-934.
doi: https://doi.org/10.12669/pjms.36.5.2379
Disclosure: None declared in the paper.
Newsletter
Like what you’re reading? Subscribe to Dermatology Times for weekly updates on therapies, innovations, and real-world practice tips.