- General Dermatology
- Eczema
- Alopecia
- Aesthetics
- Vitiligo
- COVID-19
- Actinic Keratosis
- Precision Medicine and Biologics
- Rare Disease
- Wound Care
- Rosacea
- Psoriasis
- Psoriatic Arthritis
- Atopic Dermatitis
- Melasma
- NP and PA
- Skin Cancer
- Hidradenitis Suppurativa
- Drug Watch
- Pigmentary Disorders
- Acne
- Pediatric Dermatology
- Practice Management
CP-GEP Model Successful in Stratifying Node-Negative Patients into Risk Groups for Melanoma Disease Progression
According to 2 studies, this model has demonstrated the potential to assist in developing treatment and monitoring plans for early-stage melanomas.
SkylineDx, a biotech company headquartered in Rotterdam, Netherlands, with a presence in San Diego, California, recently announced the publication of 2 scientific publications supporting the success of the company's Merlin test, or CP-GEP model, in stratifying patients with early-stage cutaneous melanoma for risk and long-term survival.1
The individual publications were revealed at the 20th European Association for Dermato-Oncology Congress, which ran from April 4 to 6 in Paris, France.
"Both studies provide promising insights into CP-GEP's capacity to stratify patients based on their risk for disease progression and its potential value to support treatment and surveillance strategies for early melanomas," said Jvalini Dwarkasing, PhD, chief scientific officer at SkylineDx, in a news release.1
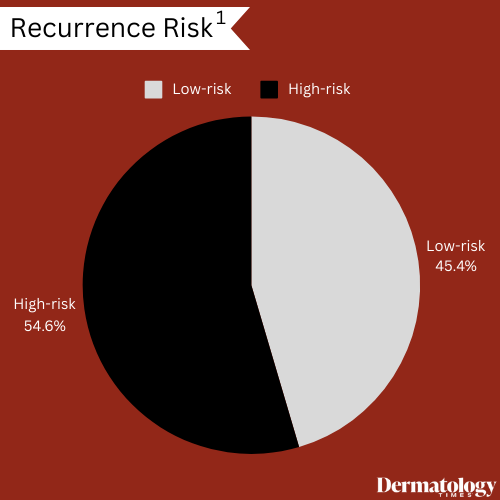
The first of the publications, a study conducted by researchers from Denmark, analyzed 438 patients diagnosed with T1-T3 cutaneous melanoma (stage I-II) treated between 2010 and 2015 at two university clinics in Denmark. Patients had undergone sentinel lymph node biopsies and were found negative for metastasis.2
Researchers collected archived tissue samples of primary melanomas and analyzed them utilizing the CP-GEP model. The primary outcome measured by researchers was the 5-year recurrence-free survival, while the secondary outcome was the 5-year overall survival.
Results demonstrated that the model effectively distinguished between low and high-risk groups for disease progression. Out of the 438 patients, CP-GEP classified 199 as low-risk and 239 as high-risk for recurrence. The low-risk group exhibited a 5-year recurrence-free survival of 92.0%, compared to 82.8% in the high-risk group. Similarly, the 5-year overall survival for the low-risk group was 92.5%, while it was 86.6% for the high-risk group. In the high-risk group, 17.2% experienced disease recurrence within 5 years compared to 8% in the low-risk group.
"CP-GEP can stratify stage I-II melanoma patients into high and low-risk for recurrence, suggesting its potential value in treatment decision-making and surveillance strategies for stage I-II melanoma," according to authors of the publication.2
In the second publication, researchers focused on assessing CP-GEP's efficacy in stratifying patients with head and neck melanoma who had not undergone sentinel lymph node biopsy based on their risk of recurrence.3
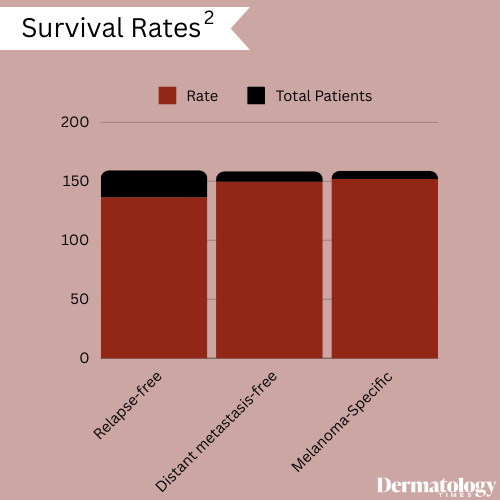
Researchers analyzed formalin-fixed paraffin-embedded primary tumor samples from 451 patients with stage I/II cutaneous melanoma who were diagnosed between 2000 and 2017, as recorded in the Central Malignant Melanoma Registry. These patients did not undergo sentinel lymph node biopsy.
The CP-GEP model utilized in the study combined the expression levels of 8 genes (SERPINE2, GDF15, ITGB3, CXCL8, LOXL4, TGFBR1, PLAT, and MLANA) determined by quantitative reverse transcription polymerase chain reaction, along with patient age and Breslow thickness, to categorize patients into CP-GEP low- or high-risk groups. The study assessed relapse-free survival, distant metastasis-free survival, and Melanoma-Specific Survival using Kaplan-Meier curves.
Among the 451 included patients, of whom had been diagnosed with stage IA-IIC melanoma, 40% were females, with a median age of 63 years and a median Breslow thickness of 0.5 mm. Of these patients, 20% (90 patients) had been diagnosed with head and neck melanoma, in particular. The interim analysis conducted on samples from 159 patients revealed promising survival rates: a 5-year relapse-free survival of 85.8%, distant metastasis-free survival of 94.1%, and Melanoma-Specific Survival of 95.7%.
"CP-GEP has the potential to risk stratify pts with early-stage melanoma that did not undergo SLNB based on their risk for long-term survival," according to the publication's authors. "Patients with CP-GEP Low-Risk have a good long term survival compared to High-Risk patients even though SLN status was not assessed, and this seems to be true also for pts with H&N melanoma. This may allow the clinicians to skip SLNB in this difficult anatomic localization."
References
- Independent European data on CP-GEP model in stratifying melanoma patients for long term survival. News release. PR Newswire. April 5, 2024. Accessed April 9, 2024. https://www.prnewswire.com/news-releases/independent-european-data-on-cp-gep-model-in-stratifying-melanoma-patients-for-long-term-survival-302109231.html#:~:text=The%20findings%20indicate%20that%20the,in%20the%20low%2Drisk%20group
- Weitemeyer MBM, Helvind NM, Klausen S, et al. Using a clinicopathologic and gene expression model to predict prognosis in stage I-II primary cutaneous melanoma: a multicenter Danish cohort study. EADO 2024. Accessed April 9, 2024. Weitemeyer EADO 2024.pdf (falconprogram.com)
- Amaral T, Chatziioannou E, Nuebling A, et al. Identification of patients at high risk for relapse by Merlin Assay (CP-GEP) in an independent cohort of melanoma patients (pts) that did not undergo sentinel lymph node biopsy: a H&N subgroup analysis. EADO 2024. Accessed April 9, 2024. Amaral EADO 2024.pdf (falconprogram.com)

